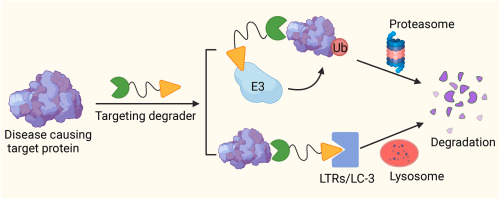
From “Inhibition Failure” to “Precision Elimination”: How Targeted Protein Degradation (TPD) 2.0 Is Reshaping the Future of Drug Development
Over the past decades, “inhibition” has been the dominant strategy in drug development—using small molecules or antibodies to bind target proteins and block their function to achieve therapeutic effects. However, as disease complexity continues to increase, traditional “occupancy-driven inhibition” has revealed significant limitations.
On one hand, many disease-causing proteins lack suitable binding pockets and are considered “undruggable targets.” On the other hand, even when successfully inhibited, the proteins remain present and may regain function through mutation or overexpression, leading to drug resistance.
Against this backdrop, Targeted Protein Degradation (TPD) has rapidly emerged and entered what is widely recognized as the “2.0 era” in 2025–2026. The core paradigm is no longer about “what a protein does,” but rather “eliminating the protein entirely.”
As a long-term observer of global innovative drug trends,
👉 DengYueMed global pharmaceutical intelligence platform
believes that TPD represents not only a technological breakthrough but also a fundamental shift in the logic of drug development.
I. A Mechanistic Revolution: From “Functional Inhibition” to “Protein Elimination”
The essence of TPD lies in harnessing the cell’s own protein degradation systems (such as the ubiquitin-proteasome system or lysosomal pathways) to selectively tag and degrade target proteins.
Compared to traditional inhibitors, TPD offers three major advantages:
- Event-driven rather than occupancy-driven
- High efficiency at low doses: a single molecule can repeatedly degrade multiple proteins
- Ability to target previously “undruggable” proteins
This mechanistic leap has made TPD one of the most disruptive directions in modern drug discovery and a key focus area continuously tracked by
👉 global pharmaceutical supply chain insights.
II. Three Core Technological Approaches: Building the TPD Ecosystem
1. PROTAC: Bifunctional Molecules Driving Industrialization
PROTAC (Proteolysis Targeting Chimera) is one of the most mature technologies in the TPD field. It consists of three components: a ligand for the target protein, a ligand for an E3 ubiquitin ligase, and a linker connecting the two.
By “bridging” the target protein and the E3 ligase, PROTAC induces ubiquitination and subsequent degradation of the target protein.
In recent years, PROTAC technology has undergone significant advancements:
- Expansion from nuclear proteins to cytoplasmic proteins
- Progress toward degrading membrane proteins and secreted proteins
- Improved structural design and pharmacokinetic properties
Notably, in prostate cancer, PROTAC degraders targeting the androgen receptor (AR) have entered clinical trials, demonstrating potential benefits for patients with drug-resistant disease.
2. Molecular Glue: A New Paradigm of “Induced Interaction”
Unlike PROTACs, Molecular Glue does not rely on a bifunctional structure. Instead, it uses a small molecule to enhance or induce interactions between a target protein and an E3 ligase, thereby triggering degradation.
This mechanism offers several advantages:
- Simpler molecular structure and improved drug-like properties
- Greater feasibility for oral administration
- Ability to uncover novel protein–protein interaction interfaces
In recent years, molecular glue targets have expanded from classical proteins such as IKZF1/3 to key proteins like GSPT1, significantly broadening the scope of degradable targets.
3. LYTAC: Expanding into Extracellular Targets
Traditional TPD approaches mainly focus on intracellular proteins, while LYTAC (Lysosome Targeting Chimera) extends degradation mechanisms to extracellular and membrane proteins.
Its core principle involves binding both the target protein and a cell surface receptor (such as CI-M6PR), directing the target protein to the lysosome for degradation.
This innovation is highly significant:
- Enables degradation of secreted proteins, cytokines, and membrane receptors
- Provides new strategies for targets beyond the reach of antibodies
- Holds strong potential in immune and metabolic diseases
III. Target Expansion: From “Classical Proteins” to “Undruggable Targets”
One of the most notable changes in the TPD 2.0 era is the rapid expansion of target space.
Previously, drug development focused primarily on proteins with well-defined binding pockets, such as kinases or receptors.
Today, TPD is unlocking a wide range of traditionally “undruggable” targets, including:
- Transcription factors (e.g., IKZF family)
- Translation regulatory proteins (e.g., GSPT1)
- Nuclear receptors (e.g., androgen receptor (AR))
Among these, the androgen receptor (AR) is a key driver in prostate cancer and has long been associated with resistance to conventional therapies.
For more oncology pipeline insights, see
👉 latest oncology innovation analysis.
Conclusion: TPD as the “Foundational Logic Reset” of Drug Development
The arrival of the TPD 2.0 era marks a fundamental shift in drug development logic—from “how to inhibit a protein” to “whether we can eliminate it entirely.”
This “protein elimination” strategy not only expands the boundaries of druggable targets but also provides new solutions for overcoming drug resistance and treating complex diseases.
As technologies such as PROTAC, molecular glue, and LYTAC continue to mature, TPD is expected to become the third major therapeutic platform, alongside small molecules and antibody drugs.
From an industry perspective, early positioning in TPD may determine competitive advantage in the next wave of pharmaceutical innovation.
👉 Visit the
DengYueMed homepage
for more global pharmaceutical intelligence and supply chain insights.