New Progress in China’s Pan-KRAS Inhibitor Development
According to the latest information from China’s Center for Drug Evaluation (CDE), the Class 1 innovative drug BGB-53038, developed by BeiGene, has received implicit approval to initiate clinical trials in China.
The study targets patients with advanced solid tumors, marking another important step in the evolution of KRAS-targeted therapies.
As highlighted by DengYueMed, a global pharmaceutical supply and medical intelligence platform, breakthroughs in pan-KRAS inhibitors may help address long-standing unmet needs in KRAS-mutated cancers.
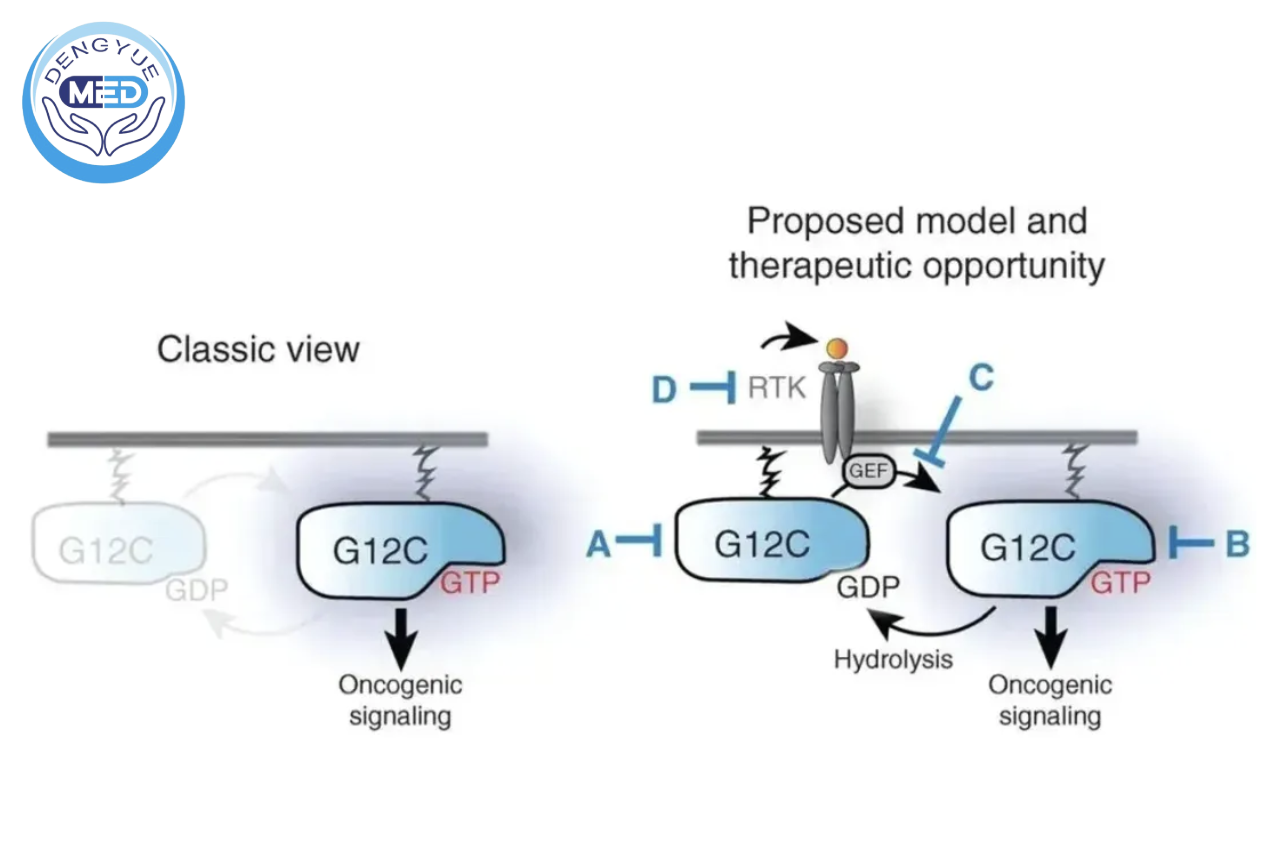
KRAS Mutations: A Longstanding Challenge
KRAS is a critical regulator in cellular signaling pathways. Once mutated, it can lead to persistent activation of oncogenic signaling.
Key facts:
- ~25% of cancer patients harbor KRAS mutations
- Major subtypes include:
- G12C
- G12D
- G12V
- G12R
These mutations are highly prevalent in:
- Non-small cell lung cancer (NSCLC)
- Pancreatic cancer
- Colorectal cancer
For decades, KRAS was considered an “undruggable” target due to the lack of clear binding pockets.
This changed in 2021 with the approval of sotorasib, the first KRAS G12C inhibitor. However, current therapies still only cover a limited subset of patients.
Pan-KRAS Strategy: Expanding Treatment Coverage
Unlike mutation-specific drugs, pan-KRAS inhibitors aim to target multiple KRAS mutations simultaneously.
Key features of BGB-53038:
1. Selective KRAS targeting
- Strong activity against KRAS
- Reduced activity on NRAS / HRAS
→ Potentially lower off-target toxicity
2. Broader clinical applicability
- Covers multiple mutation subtypes
- May benefit a wider patient population
This is particularly important in cancers with high KRAS mutation prevalence.
Clinical Development: First-in-Human Study
According to ClinicalTrials.gov (NCT06585488), BeiGene has launched a Phase I trial to evaluate:
- Safety and tolerability
- Pharmacokinetics (PK)
- Pharmacodynamics (PD)
- Preliminary efficacy
Combination strategies include:
- Tislelizumab (PD-1 inhibitor) → NSCLC
- Cetuximab (EGFR antibody) → Colorectal cancer
👉 This reflects a broader trend:
Targeted therapy + immunotherapy combinations are becoming standard.
Global Competition Intensifying
KRAS is now one of the hottest areas in oncology R&D.
Current landscape:
- ~35 pan-KRAS programs globally
- ~7 already in clinical stages
Key players include:
- Revolution Medicines
- Eli Lilly
- Jacobio Pharmaceuticals
The field is rapidly shifting from single-mutation targeting → multi-mutation strategies.
Market Potential: A Multi-Billion Opportunity
- ~2.7 million KRAS-mutant cancer patients globally per year
- G12C + G12D represent the largest segments
Estimated market potential:
👉 >$10 billion
Pan-KRAS inhibitors are widely expected to surpass single-mutation drugs in long-term value.
China’s Rising Role in Global Oncology Innovation
China’s biotech sector is rapidly moving up the value chain:
- From PD-1 → to KRAS → to next-gen platforms
- Increasing participation in global clinical trials
- More first-in-class / best-in-class strategies emerging
In this ecosystem, platforms like
DengYueMed
play a key role by:
- Tracking global innovation pipelines
- Supporting international drug access
- Bridging supply and clinical demand
Conclusion
The entry of BGB-53038 into clinical trials marks a critical step toward broad-spectrum KRAS targeting.
From “undruggable” to one of the most competitive areas in oncology, KRAS continues to reshape:
- Drug development strategies
- Clinical treatment paradigms
- Global biotech competition
As pan-KRAS therapies advance, the next phase of precision oncology will likely be defined not only by molecular innovation, but also by global execution and access capabilities.