China’s Next-Generation Antiplatelet Drug CG-0255 Shows Clinical Progress
DengYueMed, a global pharmaceutical service platform closely tracking innovative drug development, notes that cardiovascular therapeutics continue to evolve rapidly, with next-generation antiplatelet agents emerging as a key focus area.
According to publicly available information, as of March 2026, CG-0255 benzenesulfonate capsules remain under clinical development and have not yet been approved in China or globally.
The drug is developed by Shanghai Kejun Pharmaceutical Technology and represents a novel antiplatelet therapy targeting cardiovascular and cerebrovascular diseases.
👉 For more global pipeline insights:
Explore innovative drug updates
I. Clinical Development Progress
According to the Center for Drug Evaluation (CDE):
- IND application accepted: January 2025
- Phase I clinical dosing completed: March 2025
- Early results:
- Favorable safety profile
- Key endpoints met expectations
Currently:
- 🇨🇳 China: Phase I
- 🌍 Global: Some programs reportedly advancing to Phase III
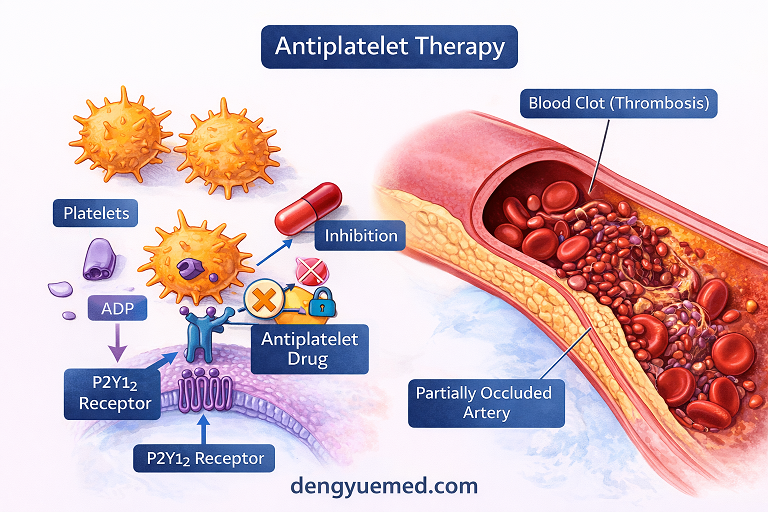
II. A Novel P2Y12 Receptor Antagonist
CG-0255 is a next-generation P2Y12 receptor inhibitor, a critical drug class in preventing arterial thrombosis.
Dual Formulation Strategy
- Intravenous injection
- Oral capsules
This enables broader use across clinical scenarios:
- Acute coronary syndrome (ACS)
- Myocardial infarction
- Ischemic stroke
- Peripheral arterial disease
Potential Advantages
- Rapid onset of action
- Favorable safety profile
- Reduced inter-patient variability
- Lower risk of drug–drug interactions
If confirmed, CG-0255 may become a competitive next-gen alternative in the P2Y12 class.
III. Global Demand for Antiplatelet Therapy
Cardiovascular disease remains a leading global cause of death.
Key facts:
- Coronary artery disease (CAD) is one of the most prevalent conditions
- Millions of deaths annually linked to thrombotic events
- Platelet aggregation = core driver of arterial thrombosis
Despite existing therapies, challenges remain:
- Bleeding risk
- Variable patient response
- Drug interactions
👉 These unmet needs are driving innovation in
global cardiovascular drug development
IV. China’s Growing Role in Innovation
China’s biopharmaceutical sector is rapidly advancing:
- Increased R&D investment
- Expansion into global clinical trials
- Focus on cardiovascular, oncology, antiviral fields
CG-0255 reflects this broader trend: 👉 From generic manufacturing → to innovative drug development
Although still early-stage, it represents a potential future option in antiplatelet therapy.
V. Final Thoughts
The development of CG-0255 highlights a key shift:
👉 Cardiovascular therapy is moving toward safer, faster-acting, and more predictable antiplatelet drugs
As clinical trials progress:
- Safety validation
- Efficacy confirmation
- Global regulatory advancement
will determine its ultimate position in the market.
DengYueMed will continue to track CG-0255 and other innovative therapies:
👉 Follow DengYueMed for latest updates
Providing timely insights into the evolving global pharmaceutical landscape.