How Do PD-1/PD-L1 Inhibitors Exert Antitumor Effects?
Against the backdrop of increasingly precise and individualized cancer treatment, immunotherapy has emerged as a major breakthrough reshaping the clinical landscape. Among these advances, PD-1/PD-L1 inhibitors are now widely used across multiple solid tumors and hematologic malignancies.
Understanding their mechanisms helps optimize treatment strategies and supports the development of combination therapies in modern oncology.
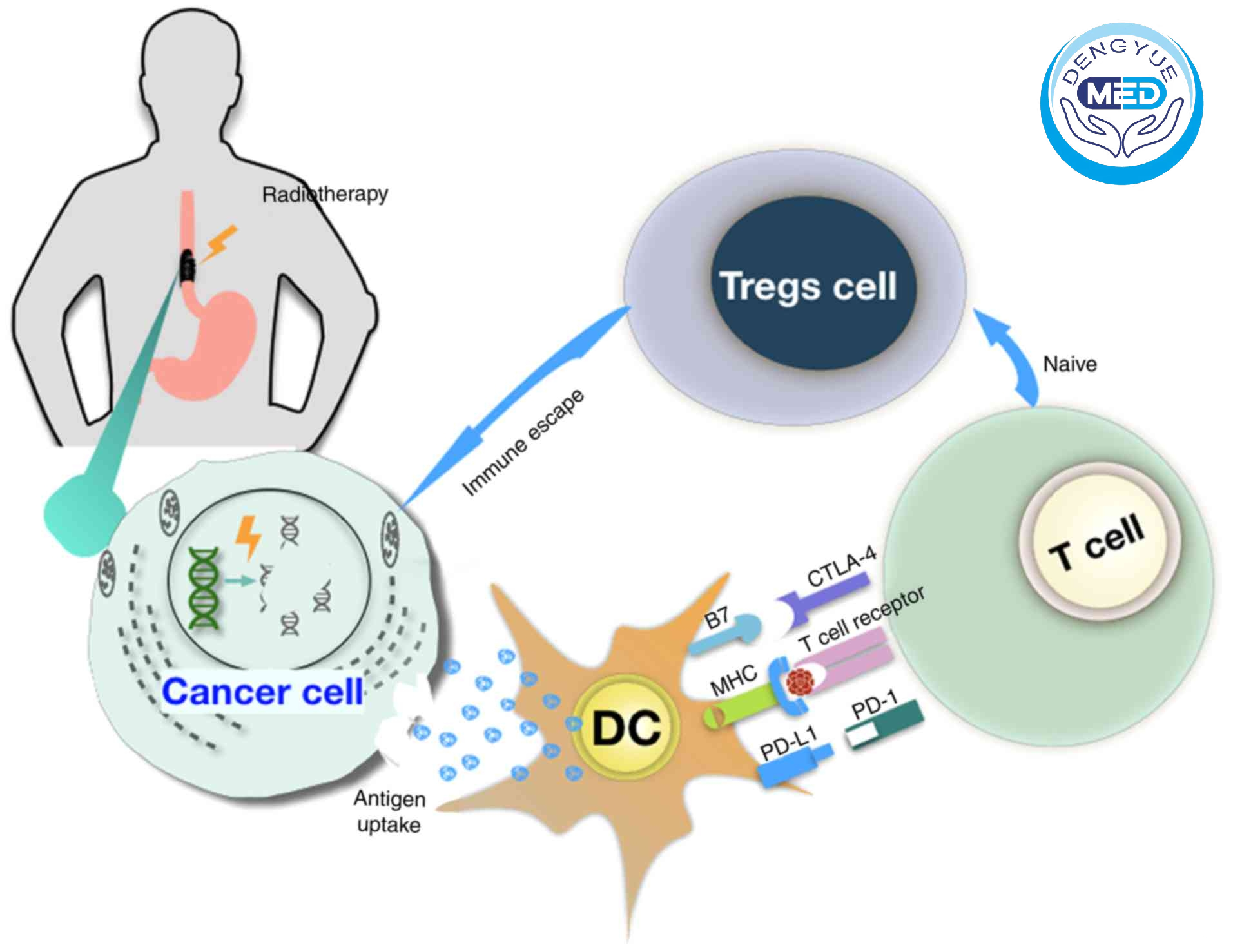
1. Immune Escape Mechanism: Why Tumors Evade the Immune System
Under normal conditions, the immune system identifies and eliminates abnormal cells through T-cell–mediated responses. However, tumor cells can suppress immune activity through immune checkpoint pathways.
A key mechanism involves:
- PD-1 receptors on T cells
- PD-L1 expression on tumor or antigen-presenting cells
When PD-1 binds to PD-L1:
- T-cell proliferation decreases
- Cytotoxic activity is suppressed
- T-cell exhaustion may occur
This allows tumor cells to survive and continue growing despite immune surveillance.
2. Mechanism of PD-1/PD-L1 Inhibitors
PD-1/PD-L1 inhibitors are monoclonal antibodies that block this inhibitory interaction.
For example, clinically used PD-1 antibodies such as
👉 Penpulimab (Anniko)
bind to the PD-1 receptor and prevent interaction with PD-L1, thereby restoring immune activity. :contentReference[oaicite:0]{index=0}
Core actions:
- Block PD-1 receptor signaling
- Prevent PD-L1 binding to T cells
Key effect:
👉 Reactivate suppressed T cells
👉 Restore immune-mediated tumor killing
Unlike chemotherapy, these drugs do not directly kill cancer cells, but instead enhance the body’s own immune response.
3. Remodeling the Tumor Microenvironment
Blocking PD-1/PD-L1 does more than activate T cells — it reshapes the entire tumor immune environment.
Effects include:
- Increased cytokine release
- Enhanced immune cell infiltration
- Improved tumor antigen exposure
This creates a positive immune feedback loop.
Additionally, immune checkpoint inhibitors may also influence immunosuppressive components such as regulatory T cells and myeloid-derived suppressor cells.
4. Clinical Application: From Monotherapy to Combination Therapy
PD-1/PD-L1 inhibitors are now widely applied in:
- Non-small cell lung cancer (NSCLC)
- Melanoma
- Cervical cancer
For instance,
👉 Enlonstobart (Enshuxing)
is a PD-1 inhibitor approved for PD-L1–positive cervical cancer, demonstrating how immune checkpoint blockade is used in real-world oncology treatment. :contentReference[oaicite:1]{index=1}
Evolution of use:
- Initially: late-line monotherapy
- Now: frontline combination therapy
Combination strategies may include chemotherapy, radiotherapy, or targeted therapy.
5. Mechanism Extension: Link to RNA-Based Therapies
While PD-1/PD-L1 inhibitors regulate immune checkpoints, newer therapies operate at the gene expression level.
For example,
👉 RNA interference therapy (Vutrisiran)
works by silencing disease-related mRNA, representing a different but complementary therapeutic strategy. :contentReference[oaicite:2]{index=2}
This highlights a broader trend:
👉 modern medicine is shifting from protein-level targeting → gene-level regulation
6. Safety and Immune-Related Adverse Events
Because these drugs activate the immune system, they may also trigger immune-related adverse events (irAEs).
Common risks:
- Skin reactions
- Thyroid dysfunction
- Hepatitis
- Pneumonitis
These effects result from immune-mediated damage to normal tissues rather than direct toxicity.
👉 Clinical management requires:
- Continuous monitoring
- Early intervention
- Individualized treatment strategies
7. Conclusion
PD-1/PD-L1 inhibitors represent a paradigm shift in oncology by:
- Blocking immune checkpoint pathways
- Restoring T-cell function
- Reshaping the tumor microenvironment
From agents like
👉 Penpulimab (Anniko)
to emerging RNA-based approaches, modern oncology is evolving toward more precise and system-level treatment strategies.
As biomarker research and combination therapies continue to advance, immunotherapy will remain a central pillar of precision medicine.