The CAR-T 2.0 Era Has Arrived
In April 2026, CAR-T cell therapy is undergoing a pivotal transformation — evolving from the “1.0 era” characterized by high cost, long production timelines, and significant toxicity, into a more clinically accessible 2.0 era.
This new phase is defined by three pillars:
Faster + Smarter + Safer
For patients, this shift signals a transition from a last-resort therapy to a more accessible and earlier-line treatment strategy.
1. Rapid Manufacturing: From Weeks to Days
A major limitation of CAR-T 1.0 was its production timeline of 2–4 weeks.
In the CAR-T 2.0 era:
- Production timelines reduced to 24–72 hours in some platforms
- Better preservation of central memory T cells (Tcm)
- Lower risk of disease progression during waiting
Some dual-target CAR-T therapies are now delivered within 10–14 days, reducing reliance on bridging therapy.
In Vivo CAR-T: A Disruptive Model
A key innovation is in vivo CAR-T, where:
- No ex vivo cell engineering is required
- CAR-T cells are generated directly inside the patient
Advantages:
- Simplified workflow
- Potential outpatient use
- Reduced cost structure
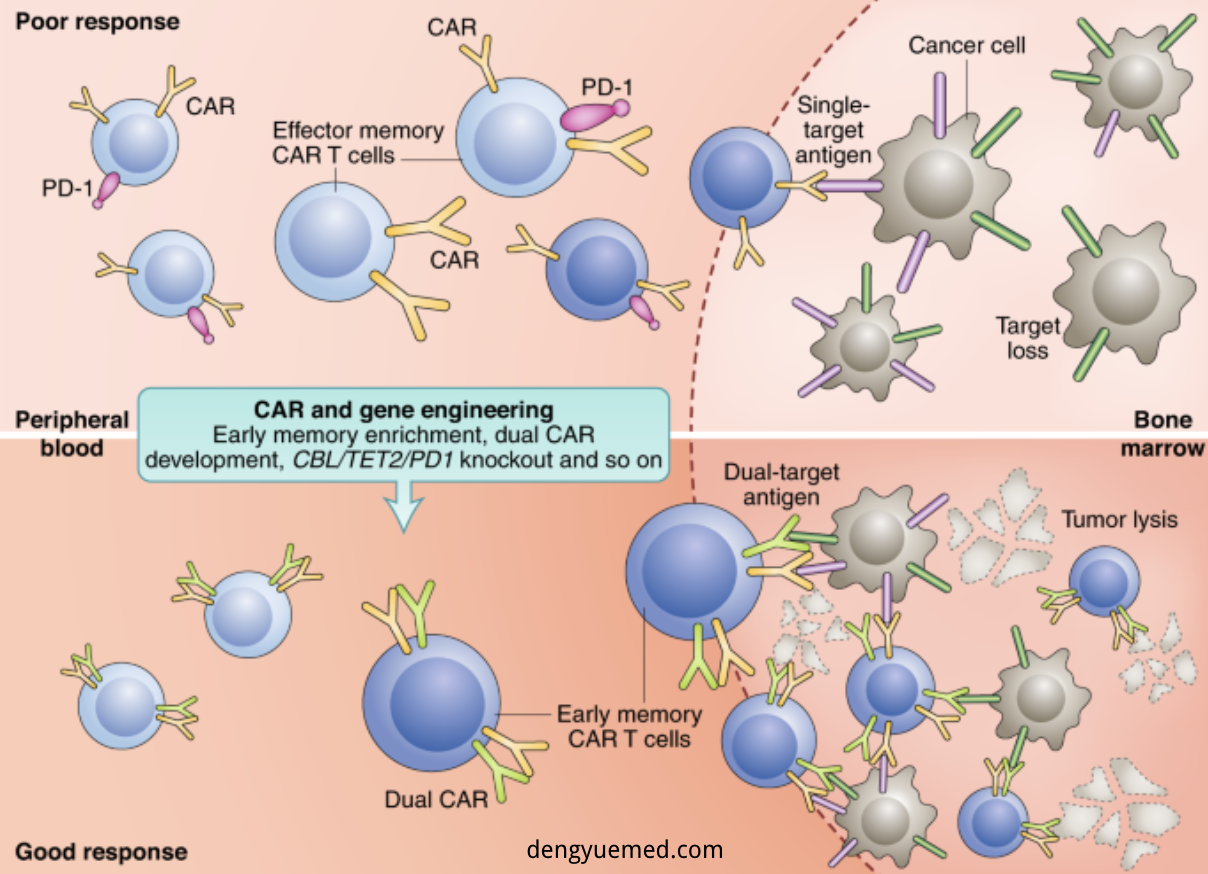
2. Dual Targeting: Overcoming Antigen Escape
Single-target CAR-T therapies often face relapse due to antigen loss.
Solution: Dual-target CAR-T
Common combinations:
- Hematologic: CD19/CD20, BCMA/CD19
- Solid tumors: CLDN18.2, GPC3
Clinical impact:
- ORR up to 93%–100% in some studies
- Reduced relapse risk
- Improved durability
Solid Tumor Breakthrough
CAR-T is expanding into:
- Gastric cancer (CLDN18.2)
- Liver cancer (GPC3)
- Pancreatic cancer
China is emerging as a major hub for these innovations.
3. Toxicity Optimization: Safer Therapies
CAR-T 1.0 was associated with:
- Cytokine Release Syndrome (CRS)
- Neurotoxicity (ICANS)
CAR-T 2.0 improves safety through:
Key strategies:
- Lower dosing with maintained efficacy
- Enhanced tumor targeting (homing)
- Armored CAR-T (cytokine support like IL-7, IL-15)
- Safety switches for emergency control
- Allogeneic CAR-T (off-the-shelf products)
Clinical improvements:
- Faster tumor response
- Reduced severe toxicity
- Less need for ICU-level management
4. Key Trends to Watch in 2026
1) Growth of Chinese Clinical Data
- Expansion in oncology and autoimmune diseases
2) Solid Tumor Expansion
- Focus on gastric, liver, and pancreatic cancers
3) In Vivo + Rapid Manufacturing Convergence
- Single-dose therapies
- AI-assisted design
4) Combination Therapies
- CAR-T + bispecific antibodies
- CAR-T + ADCs
- CAR-T + checkpoint inhibitors
5) Improved Accessibility
- Lower costs
- Faster production
- Policy support
5. From Innovation to Real-World Access
Despite progress, patients still face challenges:
- Therapy selection
- Clinical trial access
- Geographic limitations
- Risk evaluation
This highlights the importance of structured medical access pathways and informed decision-making.
6. Outlook: CAR-T as a Platform Therapy
CAR-T is evolving into a foundational therapeutic platform:
- From blood cancers → solid tumors → autoimmune diseases
- From personalized → standardized → off-the-shelf
- From single therapy → combination ecosystem
Future integration with:
- Gene editing
- AI-driven design
- Advanced delivery systems
will further expand its clinical impact.
Conclusion
CAR-T 2.0 marks a major shift in cancer treatment:
- Faster manufacturing
- Smarter targeting
- Safer profiles
What was once a last-resort therapy is now moving toward a mainstream, scalable treatment model.
The next phase of oncology will not just be about new drugs —
but about how advanced therapies are engineered, delivered, and integrated into clinical practice.