China’s ADC Global Expansion: From Licensing to Technology Leadership
As a China-based pharmaceutical distributor serving global markets, DengYueMed has been closely tracking a major industry shift:
Chinese antibody-drug conjugates (ADCs) are rapidly expanding globally, transitioning from asset out-licensing to technology-driven collaboration.
As of early 2026:
- ~44 ADC-related deals completed
- Total disclosed value exceeds $53.2 billion
- Over $3.1 billion upfront payments
More importantly, deal structures are evolving:
- Single-asset licensing → multi-asset partnerships
- Product export → platform-level collaboration
I. A Three-Phase Evolution of China’s ADC Globalization
Phase 1 (2019–2021): Late-Stage Asset Licensing
A landmark deal:
- RemeGen → Seagen (~$2.6B)
Key characteristics:
- Late-stage / near-commercial assets
- Mature clinical data
- Low risk for global partners
➡️ China acted mainly as an asset provider
Phase 2 (2022–2024): Pipeline Partnerships
A representative case:
- Kelun-Biotech × Merck (~$11B, 7 ADC assets)
Trends:
- Early-stage (Phase I/II) assets included
- Multi-target expansion (TROP2, CLDN18.2, c-MET)
- Bundled deal structures
➡️ Shift toward early innovation collaboration
Phase 3 (2025–Now): Platform-Level Collaboration
China ADC globalization enters a new phase:
- Platform licensing
- Multi-asset co-development
- Long-term strategic alliances
Examples:
- DualityBio × BioNTech
- Hansoh Pharma × GSK
➡️ Core value = technology platforms, not single assets
II. Key Drivers Behind Global Interest in Chinese ADCs
1. Full Value Chain Innovation
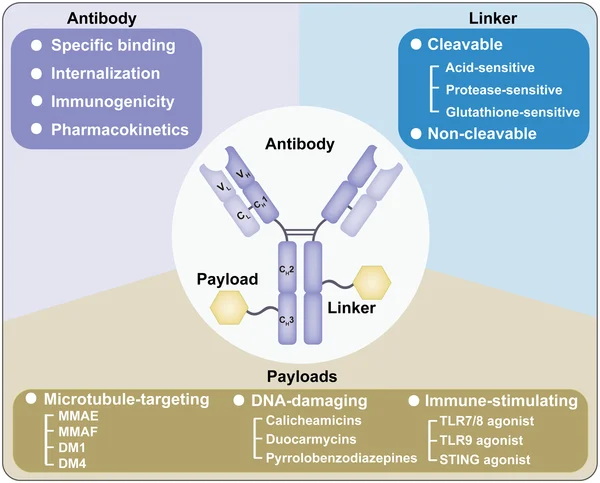
ADC development consists of:
- Antibody
- Linker
- Payload
China’s progress:
- Antibody → bispecific & multispecific
- Linker → improved stability & release control
- Payload → novel mechanisms (non-P-gp, immune modulation)
➡️ Transition: me-too → me-better → potential first-in-class
2. Differentiated Clinical Strategy
While global pipelines focus on:
- HER2
- TROP2
China is exploring:
- B7-H3
- CLDN18.2
- Novel bispecific ADCs
➡️ Focus on unmet clinical needs + differentiation
3. Clinical Development Efficiency
China offers structural advantages:
- Faster patient enrollment
- 12–24 month early validation timelines
- Lower early-stage costs
➡️ Reduced risk for global pharma partners
III. Structural Challenges and Risks
Despite rapid growth, several challenges remain:
1. Target Concentration
-
70% of deals focus on limited targets
- Risk of overcompetition & pricing pressure
2. Milestone Dependency
-
90% of deal value = milestone-based
- Upfront payments relatively low
➡️ Value realization depends on:
- Clinical success
- Regulatory approval
- Commercial performance
3. Limited Global Commercial Capability
- Most commercialization handled by multinational partners
- Chinese companies lack independent global infrastructure
4. Intellectual Property Constraints
- Linker & payload IP still dominated by global players
- Increasing risk of patent competition
IV. From Cost Advantage to Innovation Power
China’s ADC expansion reflects a broader shift:
- Cost-driven → innovation-driven
- Manufacturing advantage → R&D capability
ADC is only the beginning.
Future expansion areas include:
- Bispecific antibodies
- Cell therapies (CAR-T)
- RNA therapeutics
V. Further Reading
For a deeper analysis of ADC targets, pipelines, and approvals, see our detailed report on ADC drugs by target in 2026.
Conclusion
China’s ADC globalization is no longer about exporting individual drugs —
it is about exporting technology platforms and innovation capability.
As global collaboration deepens, Chinese biopharma is becoming:
- A core innovation contributor
- A strategic partner in global oncology development
DengYueMed will continue to track these trends and support the global distribution and accessibility of high-quality innovative therapies.