How Bispecific ADCs Overcome Antigen Escape: Technical Mechanisms and Insights
Amid the rapid expansion of the global oncology drug market, antibody–drug conjugates (ADCs) have increasingly become a cornerstone of precision cancer therapy.
The global pharmaceutical distributor DengYueMed possesses extensive experience in oncology drug supply, providing research institutions and healthcare facilities with access to the latest treatment options.
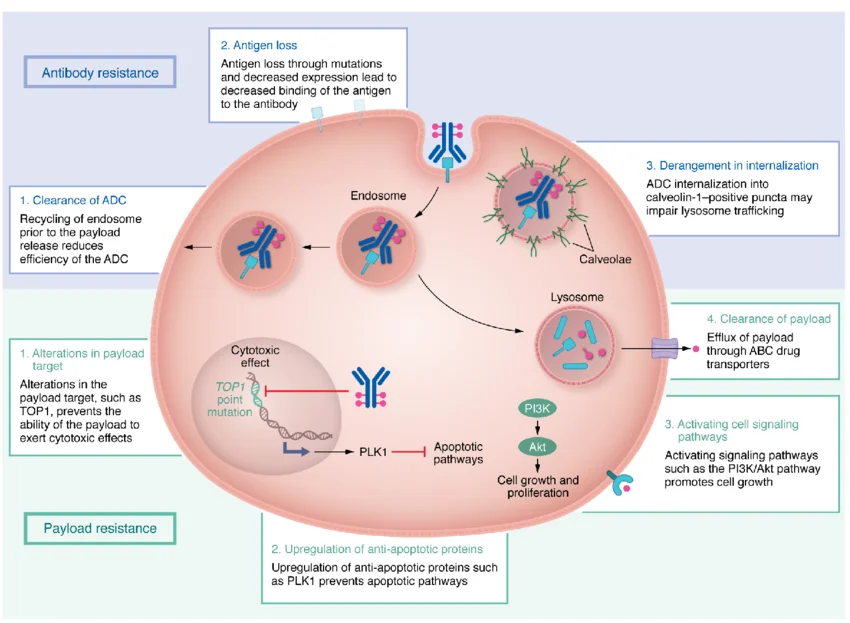
In recent years, one of the greatest challenges facing single-target ADCs has been antigen escape, which significantly limits therapeutic efficacy. Antigen escape refers to the process by which tumor cells evade ADC recognition by downregulating, losing, or mutating the target antigen, ultimately leading to treatment failure or drug resistance.
To address this issue, bispecific ADCs leverage innovative structural designs and mechanisms of action, offering a novel solution in clinical oncology.
Antigen Escape: A Key Limitation of Single-Target ADCs
Single-target ADCs rely on specific surface antigens on tumor cells to achieve targeted cytotoxicity. However, tumor cells are highly heterogeneous and can evade ADCs through multiple mechanisms:
-
Antigen downregulation or loss: Under drug pressure, tumor cells may reduce antigen expression or acquire antigen mutations, preventing ADC binding. For example, in HER2-low breast cancer patients, the efficacy of single-target HER2 ADCs is markedly reduced.
-
Tumor heterogeneity: Within a single tumor, different cell populations may exhibit variable antigen expression, meaning a single-target ADC may only eliminate a subset of cells, leaving residual tumor cells that contribute to relapse.
-
Intracellular drug trafficking limitations: Even if an ADC successfully binds its target, inefficient internalization or payload release can reduce cytotoxicity.
Antigen escape not only diminishes therapeutic response but also accelerates resistance, limiting the effectiveness of single-target ADCs in breast cancer, ovarian cancer, and hematologic malignancies.
Technical Principles and Mechanisms of Bispecific ADCs
Bispecific ADCs simultaneously target two distinct tumor antigens, broadening tumor recognition and significantly mitigating the impact of antigen escape. The core mechanisms include:
1. Dual Antigen Recognition and Synergistic Binding
Bispecific ADCs typically employ dual antibody chain structures or single-chain variable fragments (scFv) capable of binding two antigens simultaneously. For example, in breast cancer, a HER2/Trop2 bispecific ADC can target both high- and low-expression tumor cell populations, reducing treatment blind spots.
Dual-antigen engagement not only enhances avidity but also facilitates ADC internalization, increasing the efficiency of payload delivery into tumor cells.
2. Enhanced Internalization and Controlled Drug Release
Following dual-antigen binding, bispecific ADCs are internalized via receptor-mediated endocytosis and release their payloads within tumor cells or the tumor microenvironment. Modern bispecific ADCs often utilize cleavable or pH-sensitive linkers, ensuring precise drug release in acidic environments while minimizing off-target toxicity.
This design improves cytotoxic efficiency per internalization event, maintaining high efficacy even in complex tumor microenvironments.
3. Redundant Antigen Mechanism to Reduce Resistance
Single-target ADCs depend entirely on one antigen, making them vulnerable to resistance if antigen expression is downregulated. Bispecific ADCs establish redundancy: even if one target is downregulated or mutated, the other can guide the ADC into tumor cells, significantly delaying the onset of resistance.
This mechanism is particularly valuable in highly heterogeneous tumors, where single-target approaches often fail to cover all tumor subpopulations.
4. Payload and Conjugation Optimization
Bispecific ADCs employ refined drug-to-antibody ratio (DAR) control to balance efficacy and safety.
- High DAR ensures sufficient cytotoxic payload is released per internalization event but may increase systemic toxicity.
- Low DAR reduces toxicity but may compromise efficacy.
By optimizing linker chemistry and payload stability, modern bispecific ADCs achieve potent tumor cell killing while minimizing off-target effects.
Clinical Applications and Research Progress
Breast Cancer
Breast cancer represents the most active field for bispecific ADC development. HER2/Trop2 bispecific ADCs demonstrate clear advantages in targeting both high- and low-expression tumor cells.
Early clinical data show that bispecific ADCs can extend progression-free survival (PFS) in HER2-low patients, outperforming single-target ADCs. Detailed analyses of clinical strategies, target selection, and efficacy can be found in
Bispecific ADC Development in Breast Cancer: Emerging Targets and Clinical Progress.
Hematologic Malignancies
In acute myeloid leukemia (AML), CD33/CD123 bispecific ADCs can cover multiple cell subpopulations, reducing residual disease. Preclinical studies indicate enhanced cytotoxicity and reduced likelihood of resistance, providing new options for hematologic tumor treatment.
Solid Tumors and Heterogeneous Tumors
For highly heterogeneous solid tumors, such as ovarian cancer or stomach cancer, bispecific ADCs can address diverse antigen expression patterns, lowering relapse risk due to antigen escape. By selecting appropriate antigen combinations, developers can tailor ADCs to tumor type and antigen profile.
Technical Challenges in Bispecific ADC Development
-
Complex antibody design: Bispecific antibodies must maintain structural stability, balanced affinity for both targets, and low immunogenicity.
-
Payload conjugation and DAR optimization: Payload load must be precisely controlled; excessive load increases toxicity, insufficient load reduces efficacy.
-
Manufacturing requirements: Purity, stability, and batch-to-batch consistency are critical for production.
-
Clinical trial design: Trials must account for dual-antigen heterogeneity, patient stratification, and efficacy endpoints, increasing design complexity.
Despite these challenges, advances in antibody engineering, linker optimization, and payload design are steadily addressing these limitations.
Conclusion and Future Perspectives
Bispecific ADCs overcome antigen escape through dual-antigen recognition, enhanced internalization, controlled payload release, and redundant target mechanisms, offering a novel strategy for precision oncology.
They show considerable potential in breast cancer, hematologic malignancies, and highly heterogeneous solid tumors. With continued technological innovation and clinical validation, bispecific ADCs are poised to become a major force in cancer therapy.
Global pharmaceutical distributor DengYueMed continues to support healthcare institutions with access to the latest ADC therapeutics, facilitating clinical research and adoption of innovative treatments.
For a deeper understanding of clinical progress, target selection, and efficacy data for bispecific ADCs in breast cancer, please refer to
Bispecific ADC Development in Breast Cancer: Emerging Targets and Clinical Progress.