2026 Global Oncology Innovation
The Convergence of ADCs and Bispecific Antibodies: A New Paradigm in Precision Oncology
As we move through the second quarter of 2026, the global oncology landscape is witnessing a profound transformation. The traditional boundaries between different therapeutic modalities are blurring, giving rise to a new era of “design-driven” innovation. At the heart of this shift are Antibody-Drug Conjugates (ADCs) and Bispecific Antibodies (bsAbs), which are no longer just parallel tracks of development but are increasingly converging to address the most complex challenges in cancer treatment.
Based on the latest data from AACR 2026 and recent regulatory milestones from the FDA and NMPA, this analysis explores the key trends shaping the future of targeted therapies.
1. The Rise of Bispecific ADCs: Combining Precision with Potency
One of the most significant trends in 2026 is the emergence of Bispecific ADCs. By combining the dual-targeting capability of bispecific antibodies with the cytotoxic payload of ADCs, researchers are creating “guided missiles” with unprecedented precision.
Key Advantages:
- Overcoming Heterogeneity: Targeting two different antigens (e.g., EGFR/c-Met or HER2/TROP2) reduces the risk of tumor escape due to single-antigen loss.
- Enhanced Internalization: Dual-binding can trigger more efficient receptor-mediated endocytosis, delivering more payload into the cell.
- Reduced Toxicity: Improved specificity allows for lower systemic exposure, potentially widening the therapeutic window.
👉 Market Watch: Clinical data for several EGFR/HER3 and NECTIN4/TROP2 bispecific ADCs have shown promising results in refractory solid tumors, signaling a shift from “target discovery” to “architectural optimization.”
2. AACR 2026: Target Landscape and Evolution
The target landscape at AACR 2026 reveals a clear hierarchy of innovation. While established targets remain dominant, the focus has shifted toward differentiation and resistance reversal.
| Target Rank | Primary Focus in 2026 | Key Trend |
|---|---|---|
| HER2 | HER2-low & Combinations | Transition to “design-driven” innovation |
| TROP2 | Broad Solid Tumors | Post-resistance strategies & Bystander effect |
| EGFR | Bispecific Formats | Overcoming TKI resistance in NSCLC |
| B7-H3 | Pan-Solid Tumor Access | Emerging as a major next-gen ADC target |
| CLDN18.2 | Gastric & GI Cancers | Expansion from mAbs to ADCs and CAR-T |
📌 Insight: The competition is no longer about who has a HER2 ADC, but who has the best HER2 ADC for specific patient subpopulations, such as those with brain metastases or low expression levels.
3. Regulatory Dynamics: FDA and NMPA Alignment
2026 has been a landmark year for regulatory approvals, with both the FDA and NMPA showing increased support for innovative modalities.
- Accelerated Approvals: The FDA has granted several “Fast Track” and “Breakthrough Therapy” designations to next-generation ADCs targeting B7-H3 and FOLR1.
- China’s Global Impact: The NMPA has approved several domestically developed ADCs that are now being out-licensed to global pharma giants, highlighting China’s role as a powerhouse in ADC innovation.
- Harmonization: There is a growing trend toward global simultaneous filings, with companies aiming for concurrent approvals in the US, EU, and China to maximize patient access.
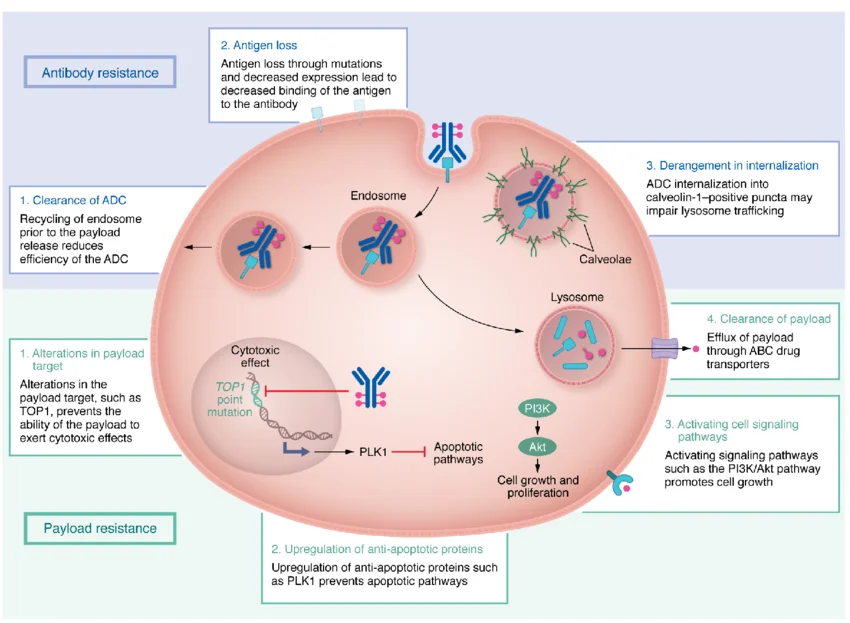
4. Challenges: The “Bottleneck” of Delivery and Resistance
Despite the excitement, the industry faces critical hurdles:
1. Payload Resistance
Tumors are developing mechanisms to pump out cytotoxic payloads. Research is now focusing on dual-payload ADCs (carrying two different toxins) to counteract this.
2. Manufacturing Complexity
Bispecific ADCs are notoriously difficult to manufacture at scale. Advances in protein engineering and site-specific conjugation are essential to ensure commercial viability.
3. Management of Adverse Events
As these drugs become more potent, managing “off-target on-tumor” toxicities (e.g., interstitial lung disease or ocular toxicity) remains a top priority for clinicians.
5. Future Outlook: Toward “Programmable” Oncology
The future of oncology lies in Programmable Medicine. We are moving toward a modular approach where antibodies, linkers, and payloads can be swapped and optimized based on a patient’s unique genetic profile.
- Immune-Synergy: Combining ADCs with PD-1/L1 inhibitors to turn “cold” tumors “hot.”
- AI-Driven Design: Using machine learning to predict the optimal antibody-antigen binding and linker stability.
📌 Conclusion: The convergence of ADCs and bispecific antibodies represents a fundamental shift in how we treat cancer—from broad-spectrum attacks to surgical-grade precision.
🌐 About DengYueMed
As a global pharmaceutical intelligence platform, DengYueMed is dedicated to tracking the most advanced innovations in oncology and rare diseases. We support global access to life-saving therapies through our expertise in regulatory policy and cross-border distribution.
👉 Explore Oncology Innovation
👉 Regulatory Updates
👉 Contact Our Experts
📩 Stay tuned for more deep dives into the technologies reshaping global healthcare.