RNA Therapeutics Trends: Commercial Breakthrough and Global Competitive Landscape in 2026

Since 2026, RNA therapeutics have continued to gain momentum, emerging as one of the most closely watched sectors in the global biopharmaceutical industry.
From siRNA to antisense oligonucleotides (ASOs), and from rare diseases to chronic conditions, RNA-based technologies are rapidly advancing toward large-scale commercialization and clinical application. A new wave of competition that could reshape the industry landscape is now fully underway.
In the Chinese market, several key developments are reinforcing this trend, including the approval of lipid-lowering siRNA therapies, innovative companies entering capital markets, continuous breakthroughs in delivery technologies, and increasing participation of domestic players in global collaborations. RNA therapeutics are approaching a true inflection point for industrialization.
DengYueMed provides a comprehensive overview of the latest developments and future trends in RNA therapeutics from the perspectives of commercialization progress, technological innovation, and global competition.
1. Accelerating Commercialization: From Proof of Concept to Blockbuster Products
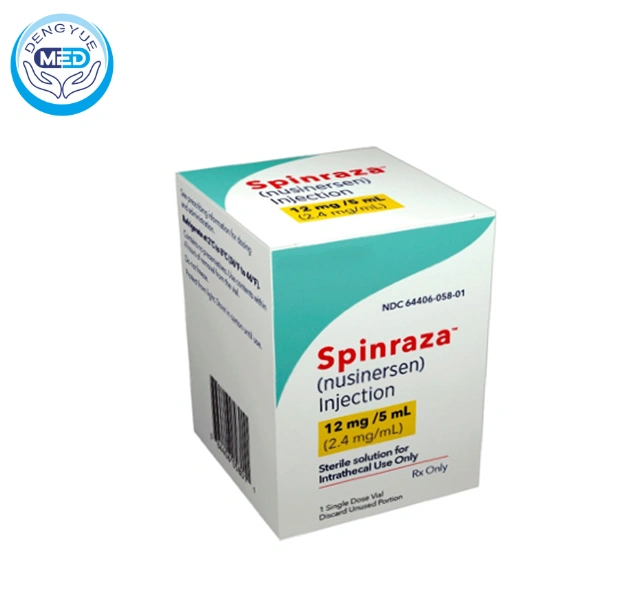
To date, more than 20 RNA therapeutics have been approved globally, covering multiple therapeutic areas such as rare diseases, cardiovascular diseases, and infectious diseases. The field has transitioned from a “proof-of-concept stage” to a “commercial harvest phase.”
In this process, Alnylam Pharmaceuticals has taken the lead in establishing a mature commercialization model. Its product Patisiran was approved in 2018 as the first siRNA-based therapy worldwide, marking the entry of RNA interference (RNAi) into clinical practice.
Building on a unified technology platform, the company has continued to expand its product portfolio, forming a well-structured pipeline:
1.Approved products span rare and metabolic diseases
2.Representative drugs include Vutrisiran, Lumasiran, and Givosiran
3.Among them, Vutrisiran surpassed USD 2 billion in sales in 2025, becoming a flagship blockbuster in the RNA therapeutics space
In the ASO field, Ionis Pharmaceuticals has also achieved steady commercial success. Its key product Nusinersen has reshaped the treatment paradigm for spinal muscular atrophy (SMA) while generating sustained revenue.
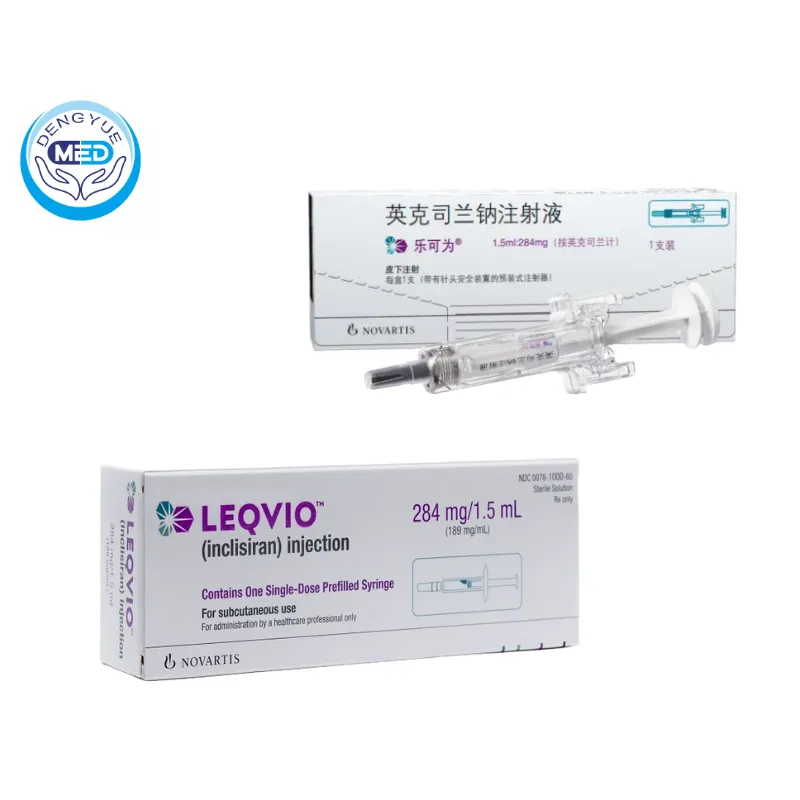
Meanwhile, Inclisiran, commercialized with the involvement of Novartis, has experienced rapid uptake in the chronic disease market. Its success is mainly driven by:
● Low dosing frequency (twice yearly), significantly improving patient adherence
● Targeting PCSK9 to deliver durable lipid-lowering effects
● Strong and sustained revenue growth, making it a benchmark for commercialization
2. From Rare Diseases to Chronic Conditions: Expanding Market Potential
Early RNA therapeutics primarily focused on rare genetic diseases, which feature well-defined mechanisms and clear targets, making them suitable for precise RNA-based interventions and benefiting from orphan drug policies.
At this stage, the key objective was to validate the technology and explore viable business models.
As the technology matures, the field is rapidly expanding into chronic diseases. A critical turning point has been the breakthrough in delivery technologies.
GalNAc-conjugated delivery systems, in particular, enable efficient targeting of hepatocytes, unlocking the vast market of cardiovascular and metabolic diseases. Key impacts include:
● Transforming the liver from a previously “hard-to-target” organ into a primary site for systemic RNA therapeutics
● Supporting drug development against key targets such as PCSK9 and APOC3
● Significantly improving molecular stability and delivery efficiency
Products such as Inclisiran, with twice-yearly dosing, are reshaping the treatment paradigm for chronic diseases.
In addition, multiple pipelines targeting key molecules are advancing, including:
● Lipoprotein(a) [Lp(a)]
● Angiotensinogen (AGT)
● Coagulation Factor XI (FXI)
Together, these developments indicate a clear trend: RNA therapeutics are becoming an important tool for long-term management of chronic diseases.
👉For a deeper understanding of RNA therapeutics in cardiovascular diseases and key targets such as PCSK9, Lp(a), and AGT, see RNA Based Therapeutic Approaches for Cardiovascular Disease: Key Targets and Clinical Advances.
3. Delivery Technology Competition: Extrahepatic Targets as the Next Frontier
While liver-targeted delivery has become relatively mature, the next phase of competition is shifting toward extrahepatic tissues, particularly the central nervous system (CNS), heart, and muscle—areas with higher technical barriers and significant unmet medical needs.
Globally, companies are pursuing multiple strategies in this space:
● Novartis continues to expand its presence in neuromuscular diseases
● Arrowhead Pharmaceuticals is advancing multi-tissue delivery platforms
● Many companies are attempting to overcome key barriers such as the blood–brain barrier
At the same time, Chinese companies are accelerating their efforts and seeking breakthroughs, such as developing cardiac-targeted delivery platforms, signaling progress toward high-barrier innovations.
4. Global Competition: The Rise of Chinese Biopharma

Amid rapid global growth, Chinese biotech companies are transitioning from followers to co-competitors, demonstrating competitiveness across multiple dimensions.
In terms of international collaboration, Chinese companies are increasingly forming strategic partnerships with multinational pharmaceutical companies, such as Eli Lilly and Roche. These collaborations typically feature:
● Large deal values, reflecting global recognition of technological value
● Coverage of global development and commercialization rights
● Acceleration of Chinese pipelines entering international markets
On the capital and industrial integration front, activities are also increasing. Companies such as Sino Biopharmaceutical are entering the RNA therapeutics field through acquisitions, signaling a transition from traditional pharma to cutting-edge technologies.
In terms of R&D strategies, Chinese companies are showing differentiated approaches:
● Early entry into major indications such as cardiovascular diseases and hepatitis B
● Focus on targets including PCSK9, ANGPTL3, and HBV
● Expansion into frontier areas such as CNS and renal diseases
5. Future Outlook: Competition in Next-Generation Therapeutic Modalities
Based on current trends, RNA therapeutics are increasingly establishing themselves as a third major therapeutic modality. Future competition will center on several key dimensions:
● Delivery technologies: breakthroughs in extrahepatic delivery will be critical to achieving leadership
● Market expansion: chronic diseases will represent the largest growth opportunity
● Global capabilities: whether Chinese companies can sustain innovation on a global scale
Conclusion
From the historic breakthrough of Patisiran to the commercial success of Inclisiran in chronic diseases, and the rapid rise of Chinese biotech companies, RNA therapeutics are entering a true “inflection point.”
Looking ahead, over the next decade, RNA-based therapies are expected to evolve from innovative alternatives to mainstream treatment modalities, continuously reshaping the global pharmaceutical landscape.
👉For a systematic understanding of RNA therapeutics in cardiovascular diseases and key target development, please refer to RNA Based Therapeutic Approaches for Cardiovascular Disease: Key Targets and Clinical Advances.
As a key connector of global innovative drug resources, DengYueMed continues to expand its presence in RNA therapeutics and high-end innovative medicines, focusing on cross-border pharmaceutical distribution, international supply chain integration, and access to rare and specialty drugs, serving global clients and partners.