China Approves New Indication for Herombopag in Severe Aplastic Anemia
As China’s innovative drug research and development capabilities continue to advance, an increasing number of medicines with independent intellectual property are entering the global spotlight.
Recently, another important development has emerged from China’s pharmaceutical industry. The Chinese pharmaceutical wholesaler DengYueMed has observed that innovative drugs developed in China are continuing to make progress in the treatment of hematologic diseases.
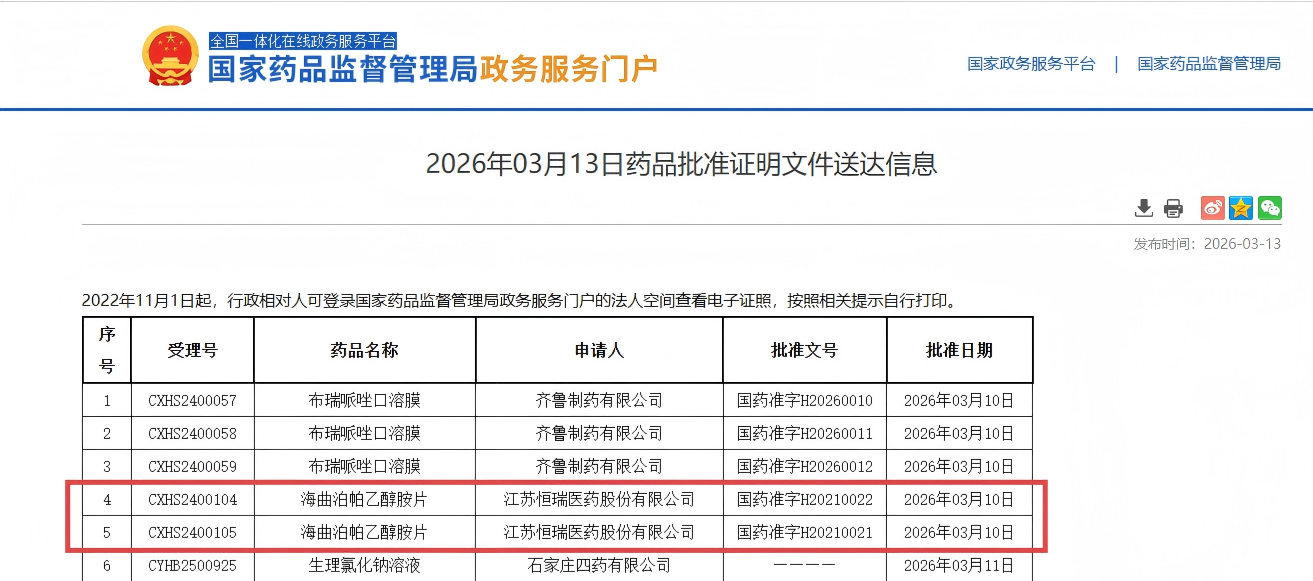
On March 13, 2026, information published on the official website of the National Medical Products Administration (NMPA) showed that Jiangsu Hengrui Pharmaceuticals Co., Ltd. has received approval for a new indication for its Class 1 innovative drug Herombopag Olamine Tablets.
The drug is now approved for use in combination with standard immunosuppressive therapy for patients aged 15 years and older with newly diagnosed Severe Aplastic Anemia (SAA).
This marks the third approved indication for the drug and represents the first time it has been authorized for first-line combination therapy for newly diagnosed SAA.
What Is Severe Aplastic Anemia?
Aplastic anemia is a serious bone marrow failure disorder often associated with immune-mediated damage to hematopoietic stem cells.
The condition is characterized by a significant decline in bone marrow hematopoietic function, resulting in reduced production of:
- Red blood cells
- White blood cells
- Platelets
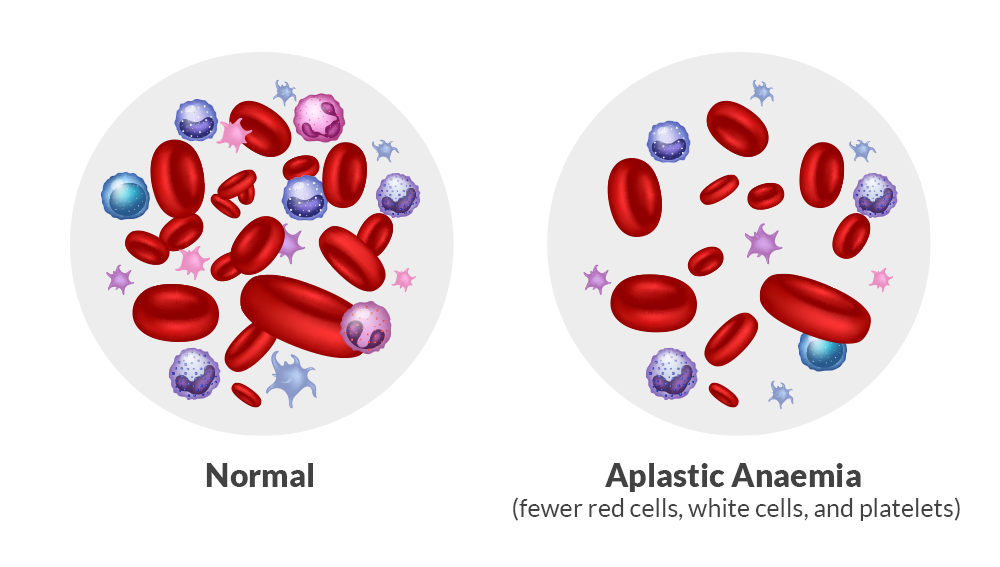
When the disease progresses to a more severe stage, it is referred to as Severe Aplastic Anemia (SAA).
Without timely treatment, SAA can lead to:
- Severe infections
- Persistent bleeding
- Complications related to anemia
- Potentially life-threatening outcomes
Epidemiological studies suggest that the incidence of aplastic anemia is generally higher in Asia than in Western countries, with particularly notable prevalence in:
- China
- Japan
- Southeast Asia
Current Treatment Strategies for SAA
According to current international clinical guidelines, the primary treatment options include:
1. Hematopoietic Stem Cell Transplantation (HSCT)
This treatment is generally recommended for younger patients with a suitable donor.
2. Immunosuppressive Therapy (IST)
A commonly used regimen includes:
- Anti-thymocyte globulin (ATG)
- Cyclosporine (CsA)
However, the complete response rate of traditional IST remains limited, and many patients respond inadequately to treatment.
Because of this limitation, therapies that stimulate hematopoiesis have become an important focus of clinical research.
Phase III Clinical Trial Demonstrates Efficacy
The approval of the new indication was primarily based on the HR-TPO-SAA-III Phase III clinical trial.
Study design:
- Multicenter
- Randomized
- Double-blind
- Placebo-controlled
- Phase III clinical trial
The study evaluated the efficacy and safety of herombopag combined with standard immunosuppressive therapy in newly diagnosed SAA patients.
The results were presented at the American Society of Hematology Annual Meeting (ASH 2024).
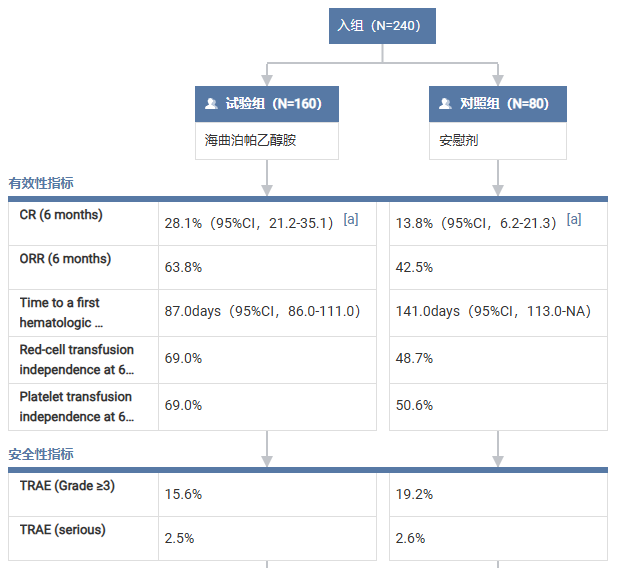
Efficacy Results at 3 Months
- Complete response (CR): 8.8% vs 5.0%
- Overall response (OR): 50.6% vs 25.0%
- Independence from RBC transfusion: 56.8% vs 27.6%
- Independence from platelet transfusion: 54.4% vs 31.2%
Efficacy Results at 6 Months
- CR: 28.1% vs 13.8% (p = 0.0129)
- OR: 63.8% vs 42.5%
- RBC transfusion independence: 69.0% vs 48.7%
- Platelet transfusion independence: 69.0% vs 50.6%
Median time to first hematologic response:
- 87 days in the herombopag group
- 141 days in the control group
These findings suggest that adding herombopag to standard therapy significantly improves treatment response and accelerates hematologic recovery.
In terms of safety, long-term treatment was generally well tolerated, and no new safety signals were observed.
Mechanism of Action of Herombopag
Herombopag olamine is an oral, non-peptide thrombopoietin receptor agonist (TPO-RA).
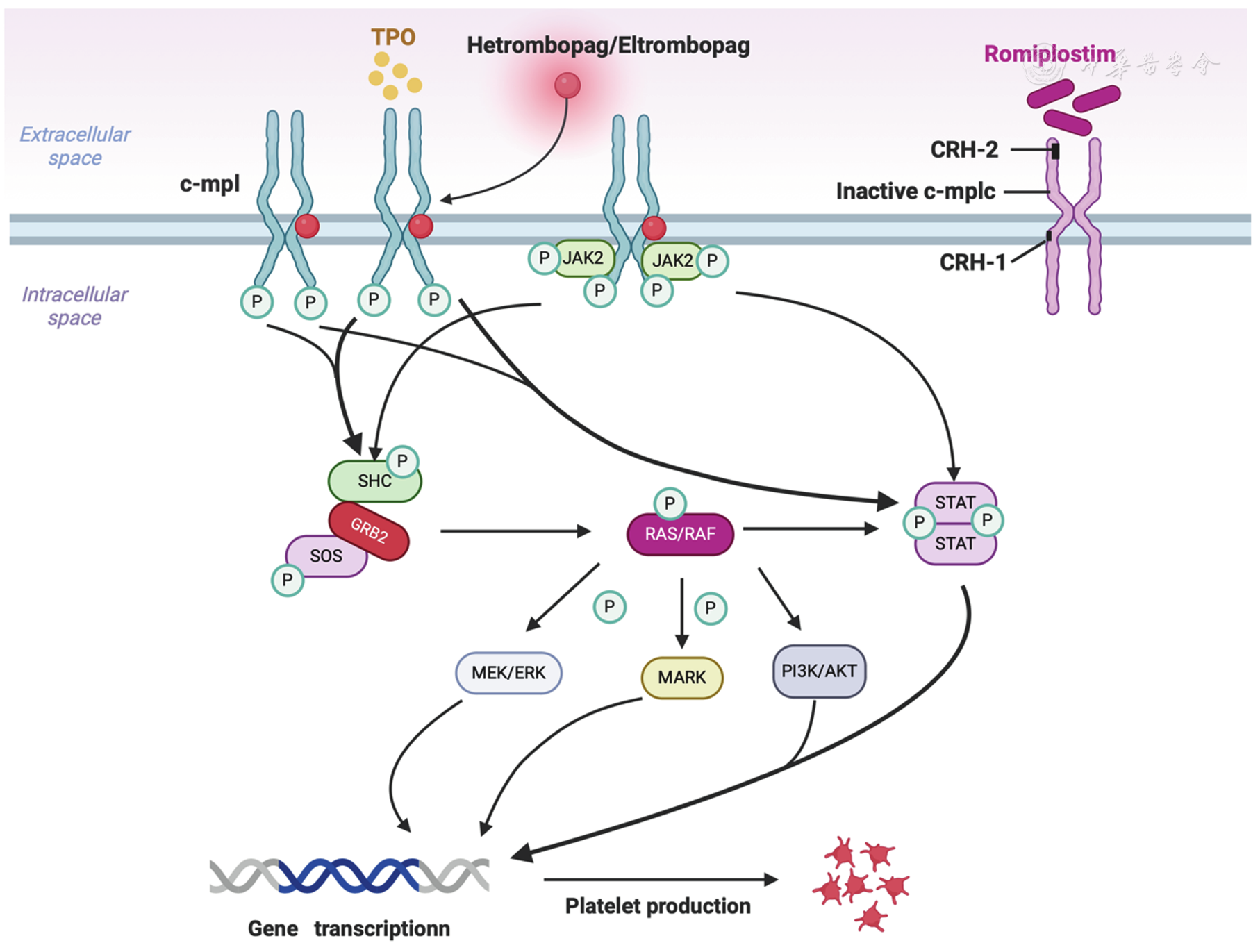
The drug works by:
- Mimicking endogenous thrombopoietin (TPO)
- Activating the TPO receptor (c-Mpl)
- Triggering downstream signaling pathways such as:
- STAT signaling pathway
- MAPK signaling pathway
These pathways promote the proliferation and maturation of megakaryocytes, increasing platelet production and improving bone marrow hematopoiesis.
Compared with traditional immunosuppressive therapy, drugs in this class offer several advantages:
- Direct stimulation of hematopoiesis
- Oral administration
- Strong combination therapy potential
As a result, TPO receptor agonists are becoming an important strategy in aplastic anemia treatment.
Approved and Ongoing Indications
Before this latest approval, Herombopag had already been approved in China for:
1. Chronic Primary Immune Thrombocytopenia (ITP)
Used in adult patients who responded poorly to treatments such as:
- Corticosteroids
- Immunoglobulin
2. Severe Aplastic Anemia
For patients who showed inadequate response to immunosuppressive therapy.
The new approval expands its use to first-line therapy for newly diagnosed SAA.
Additional indications under clinical investigation include:
- Primary immune thrombocytopenia in adults and children aged ≥6 years
- Chemotherapy-induced thrombocytopenia (CIT)
If approved, these could significantly expand the drug’s clinical applications.

The Growing Global Impact of China’s Innovative Medicines
Over the past decade, China’s pharmaceutical industry has gradually shifted from a generics-driven model to one increasingly focused on innovation.
Chinese pharmaceutical companies such as Hengrui Pharma have introduced innovative therapies across:
- Oncology
- Immunology
- Hematology
As regulatory systems evolve, Chinese innovative drugs are gaining increasing visibility at:
- International medical conferences
- Academic publications
- Global pharmaceutical markets
For physicians, researchers, and healthcare institutions worldwide, tracking the development of China’s pharmaceutical innovation is becoming increasingly important.
DengYueMed and Global Access to Chinese Medicines
DengYueMed closely follows developments in China’s pharmaceutical industry and regularly shares updates on:
- Innovative drug approvals
- Clinical research progress
- Global pharmaceutical market trends
The company also collaborates with multiple Chinese pharmaceutical manufacturers and supply networks to help international healthcare institutions obtain information on accessing innovative medicines developed in China.
Conclusion
With the emergence of more innovative therapies, China’s pharmaceutical sector is gradually evolving from a global manufacturing base into an increasingly important source of medical innovation.
As Chinese innovative medicines expand globally, companies that bridge international distribution, regulatory compliance, and pharmaceutical supply chains will play an increasingly important role in bringing these therapies to patients worldwide.