Why Humanity Continues to Invest in Orphan Drug Research
Despite Only 5% of Rare Diseases Being Treatable
Globally, thousands of rare diseases have been identified, yet only about 5% currently have approved treatments.
At first glance, this statistic raises a difficult question:
If the probability of therapeutic success is so limited, why does humanity continue to devote substantial financial, scientific, and institutional resources to orphan drug research?
Across the European Union, the United States, and Asia, governments are expanding rare disease strategies, strengthening regulatory incentives, and accelerating approval pathways. These actions signal a structural shift:
Rare disease research is no longer peripheral.
It is becoming an essential pillar of modern public health systems.
Why Has Rare Disease Become a Global Public Issue?
Although each rare disease affects a small population, collectively they impact hundreds of millions of people worldwide.
Their broader societal significance stems from several defining characteristics:
- Early onset and severe progression — Many rare diseases manifest in childhood and result in lifelong impairment
- Diagnostic complexity — Patients often face prolonged diagnostic journeys and fragmented care
- Limited therapeutic options — Most conditions rely solely on supportive or symptomatic treatment
Rare diseases therefore represent more than clinical challenges.
They are issues of equity, ethics, and social responsibility.
When life-saving needs cannot be addressed by market forces alone, public systems must intervene.
Why Continue Investing When Only 5% Are Treatable?
From a traditional commercial perspective, orphan drugs were historically considered high-risk and low-return investments:
- Small patient populations
- Difficulty recruiting for clinical trials
- Long and costly development cycles
However, over the past two decades, this perception has evolved dramatically.
1️⃣ Rare Diseases as the Origin of Scientific Breakthroughs
Many transformative therapeutic technologies were first validated in rare diseases before expanding into oncology, immunology, and broader genetic disorders.
Representative examples include:
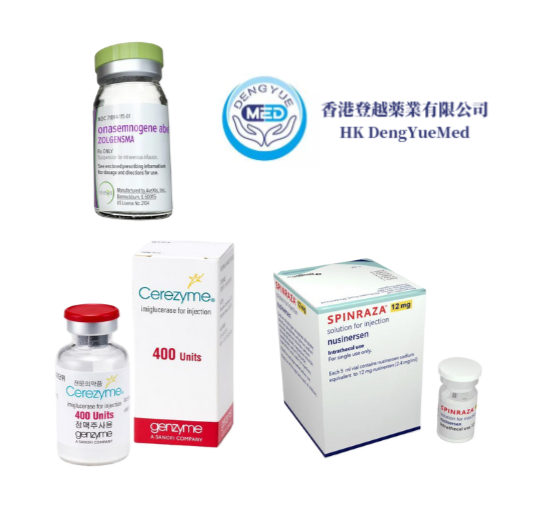
- Zolgensma — Gene therapy for spinal muscular atrophy (SMA)
- Spinraza — RNA-based therapy for SMA
- Cerezyme — Enzyme replacement therapy for Gaucher disease
These innovations did not merely treat symptoms.
They altered the natural history of disease and redefined what was medically possible.
Because many rare diseases have clearly defined genetic mechanisms, they provide ideal environments for validating precision medicine technologies.
In many cases, rare disease research becomes the proving ground for future mainstream therapies.
2️⃣ Public Health Ethics as a Driver of Policy
Rare disease patients are disproportionately children and individuals with severe genetic disorders. Their unmet needs are urgent and cannot be resolved through pure market logic.
In response, governments have introduced structural mechanisms including:
- Market exclusivity periods
- Tax credits and R&D incentives
- Accelerated regulatory review
- Dedicated funding frameworks
These policies reflect a foundational principle:
Healthcare allocation should not be determined solely by patient volume.
It must also reflect the intrinsic dignity and value of human life.
3️⃣ The Clinical Testing Ground of the Biotech Revolution
High-barrier technologies frequently complete early clinical validation in rare diseases:
- Gene editing
- Cell therapy
- Targeted molecular engineering
- Advanced molecular diagnostics
Once safety and efficacy are established in small, well-defined populations, these platforms expand to more prevalent conditions.
In this sense, sustained investment in the “5%” builds the technological and regulatory infrastructure for the remaining 95%.
The Broader Public Value of Orphan Drug Development
Transforming Individual Life Trajectories
For families affected by rare diseases, an approved therapy may mean:
- From “no available treatment” to “disease management”
- From repeated hospitalization to reintegration into education and society
- From severely shortened life expectancy to long-term survival
Such transformations extend beyond economic measurement.
They concern dignity, autonomy, and hope.
Strengthening Healthcare Systems
Rare disease initiatives have catalyzed:
- Expansion of genetic testing capabilities
- Development of specialized diagnostic networks
- Cross-border research collaboration
- More adaptive regulatory frameworks
Technological pathways initially designed for small populations often become foundational infrastructure for modern medicine.
Advancing Global Pharmaceutical Collaboration
Because rare disease patients are geographically dispersed, orphan drug development is inherently international.
Clinical trials, regulatory communication, and distribution systems must operate across jurisdictions.
In this environment, reliable and compliant supply chain networks are critical to ensuring that medical breakthroughs translate into real-world accessibility.
DengYueMed, as a global pharmaceutical distributor engaged in rare disease drug supply and service networks, focuses on bridging innovation and patient access — transforming therapeutic advances into tangible availability.
Conclusion: Measuring Compassion in Medicine
The compassion of a medical era is not measured solely by how many common diseases it cures, but by its willingness to persist for the fewest among us.
Although only 5% of rare diseases currently have treatments, humanity continues to invest — not purely out of scientific ambition, but out of commitment to fairness, dignity, and collective progress.
Orphan drugs may not immediately change the outcome of every disease.
But they represent one of the clearest expressions of modern medicine’s ethical promise:
Even when the number is small, the search for answers must continue.