CRISPR Gene Therapy Enters the Clinical Era: Casgevy Approval Marks the Dawn of Gene Editing Treatments
In recent years, gene therapy technologies have advanced rapidly. Among them, CRISPR-Cas9 gene editing has emerged as a next-generation precision medicine tool, transitioning from laboratory research into clinical application. In 2023, a landmark milestone was achieved—the world’s first CRISPR-derived therapy, Casgevy, received regulatory approval, officially marking the beginning of the “gene editing therapy era.”
This breakthrough not only reshapes the treatment paradigm for genetic diseases but also opens new possibilities for cancer, rare diseases, and even chronic conditions.
A Milestone Achievement: First CRISPR Therapy Approved
In 2023, Casgevy was approved by both the UK Medicines and Healthcare products Regulatory Agency (MHRA) and the U.S. Food and Drug Administration (FDA) for the treatment of two severe genetic blood disorders:
- Sickle cell disease
- β-thalassemia
In December 2023, the FDA approved Casgevy for patients aged 12 years and older with sickle cell disease who experience recurrent vaso-occlusive crises. This marks the first CRISPR-based gene editing therapy approved in the United States. Subsequently, its indication was expanded to include transfusion-dependent β-thalassemia, further broadening its clinical application.
Importantly, this therapy is considered a one-time treatment, with the potential to fundamentally alter disease progression rather than merely alleviating symptoms.
Mechanism of Action: From Symptom Management to Gene Correction
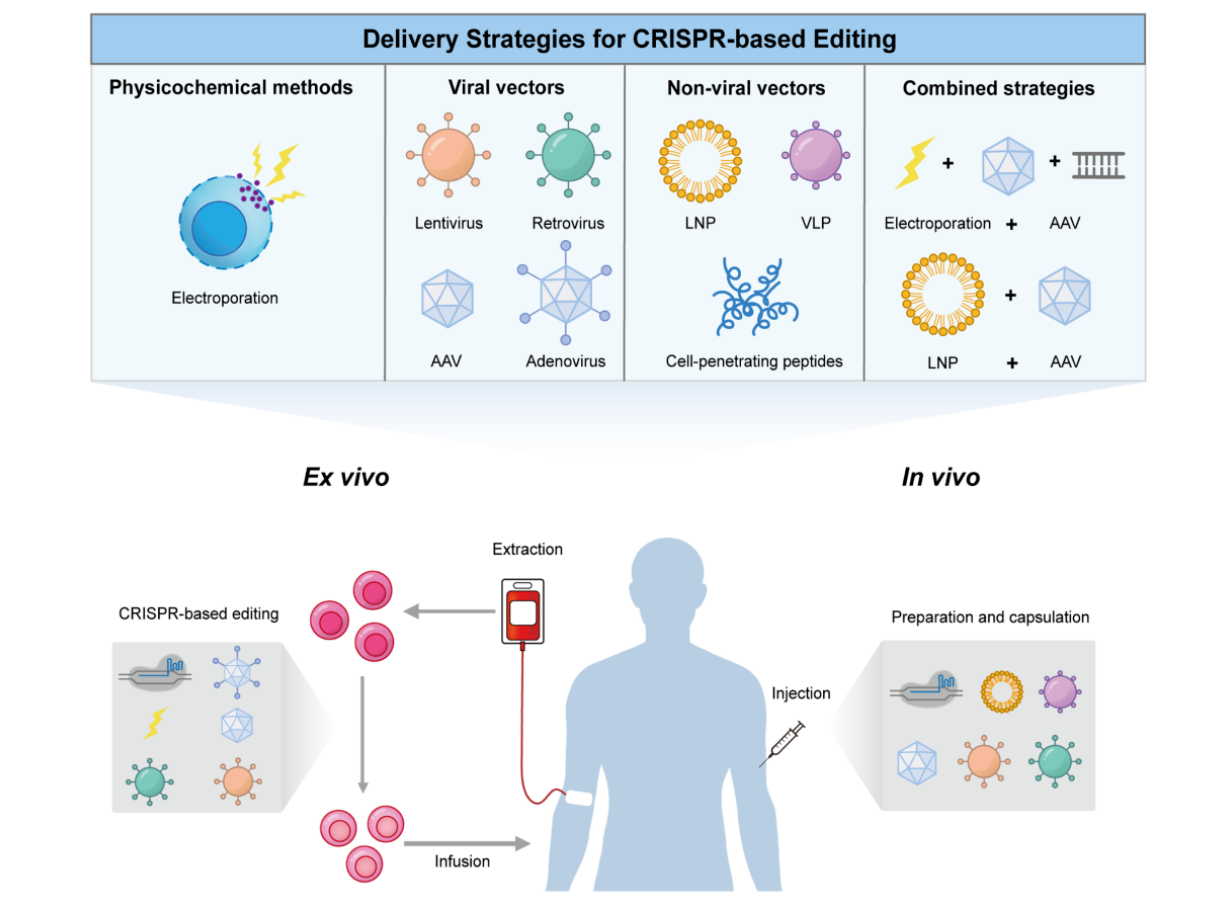
The core of Casgevy lies in using CRISPR-Cas9 technology to edit a patient’s own hematopoietic stem cells.
The general process includes:
- Collecting hematopoietic stem cells from the patient
- Editing the cells ex vivo using CRISPR technology
- Modifying gene expression (e.g., reactivating fetal hemoglobin production)
- Reinfusing the engineered cells back into the patient
Specifically, this therapy works by editing regulatory genes to increase the production of fetal hemoglobin (HbF), which can replace defective adult hemoglobin. This reduces red blood cell deformation and vascular blockage.
This mechanism introduces a fundamental shift:
- Eliminates reliance on lifelong transfusions or medications
- Transitions from “disease control” to “functional cure”
- Directly targets the genetic root cause of disease
Clinical Outcomes: Toward a Functional Cure
Clinical trial results demonstrate the transformative potential of Casgevy:
- Most sickle cell disease patients experienced a significant reduction or complete elimination of pain crises
- Many β-thalassemia patients became independent of regular blood transfusions
- Some studies report long-term sustained remission
CRISPR therapy has been shown to significantly increase fetal hemoglobin levels, thereby reducing vascular occlusion and related complications. For diseases that previously required lifelong management, this represents a shift toward near-curative treatment.
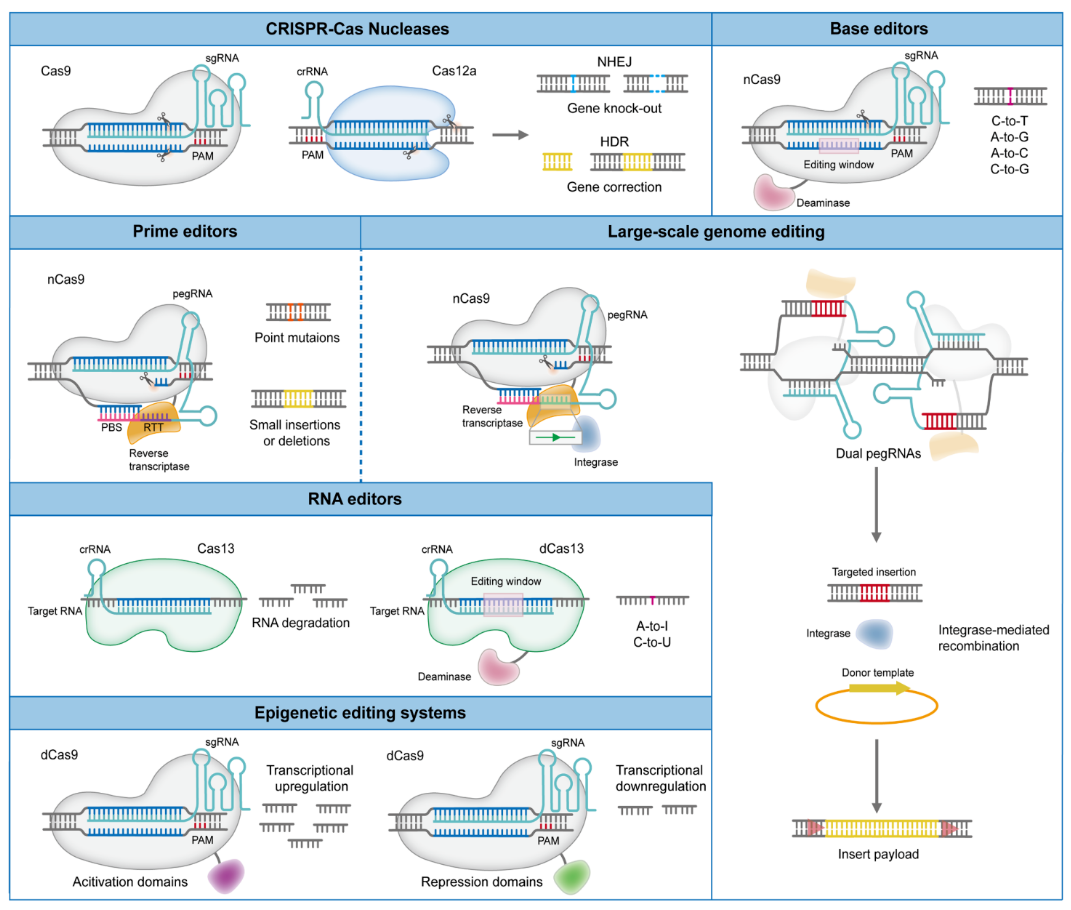
Technological Significance: Entering the Era of Precision Gene Editing
The approval of Casgevy represents more than a single product success—it signals a transformation in therapeutic paradigms. Traditional gene therapies (such as viral vector-based approaches) mainly focus on gene addition, whereas CRISPR enables:
- ✂️ Precise DNA cutting and repair
- 🎯 Higher targeting accuracy and efficiency
- 🔬 Broader applicability across disease types
This advancement has far-reaching implications:
- Providing universal solutions for genetic disorders
- Accelerating the development of personalized medicine
- Offering new tools for cancer immunotherapy
Looking ahead, CRISPR technology may also be applied to:
- Engineering immune cells (e.g., optimizing CAR-T therapies)
- Treating viral infections such as HIV
- Addressing neurological disorders
Challenges and Limitations: Barriers to Widespread Adoption
Despite its promise, CRISPR therapy still faces several practical challenges:
- High Cost: Current treatments may cost up to millions of dollars, limiting accessibility.
- Complex Treatment Process: Including stem cell collection, intensive preconditioning chemotherapy, and specialized clinical infrastructure.
- Safety Concerns: Although improving, key risks remain, such as off-target effects, long-term genomic stability, and potential tumorigenicity.
- Limited Accessibility: Currently available only in select countries and specialized centers, making global access challenging.
Industry Outlook: What Lies Ahead for CRISPR Therapies?
The success of Casgevy marks CRISPR’s transition from a research tool to a clinical therapeutic platform. Future trends include:
- 📈 More CRISPR-based therapies entering clinical trials
- 🧬 Integration with cell therapies (e.g., CAR-T, TCR-T)
- ⚙️ Technological advancements (e.g., RNA therapy)
- 🌍 Gradual improvement of global regulatory frameworks
At the same time, the global pharmaceutical supply ecosystem is evolving. Platforms like Dengyue Pharma are playing a key role by integrating international medical resources and compliant channels, helping improve patient access to cutting-edge therapies through cross-border pharmaceutical distribution.
Conclusion
The approval of Casgevy demonstrates that CRISPR gene editing has officially entered the clinical era. This milestone represents not only a technological leap but also a fundamental shift in how genetic diseases are treated.
As technologies mature, costs decrease, and healthcare systems evolve, CRISPR therapies are expected to move from breakthrough innovation to widespread clinical use—ushering in a new era of truly curative medicine.