The Rise of China’s Rare Disease Drugs
From Follower to Emerging Global Leader
Executive Summary
China’s rare disease pharmaceutical sector is undergoing structural transformation.
What was once a generics-dominated industry is evolving into an innovation-capable ecosystem increasingly integrated into the global orphan drug landscape.
Regulatory acceleration pathways, domestic R&D capability upgrades, pricing advantages, and industrial integration are collectively reshaping China’s position in rare disease therapeutics.
The shift is not incremental. It is systemic.
Rare diseases may be “rare” individually, but collectively they affect hundreds of millions of patients worldwide.
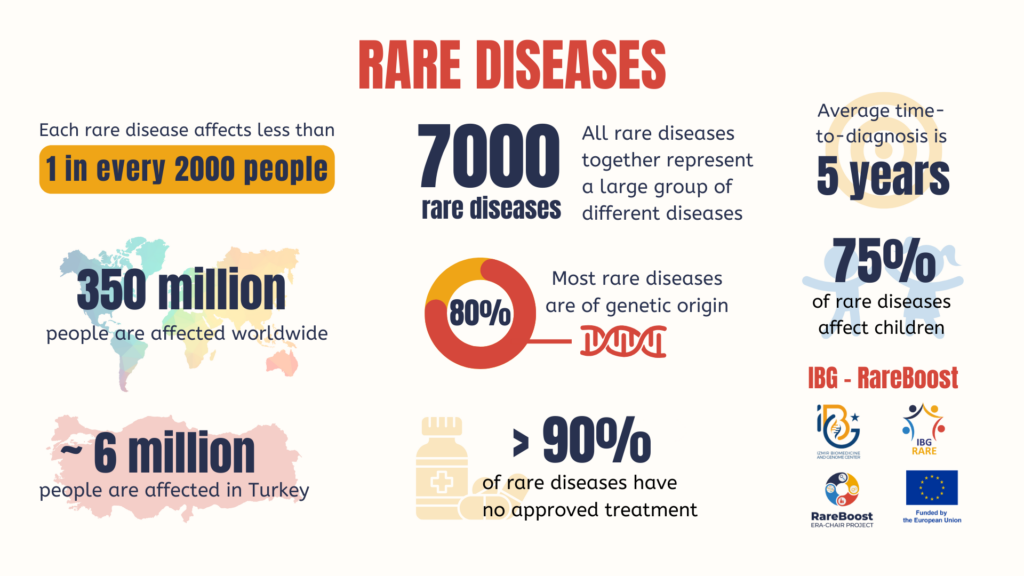
Figure 1. Over 7,000 rare diseases affect an estimated 300–400 million people globally.
Historically, the orphan drug market was largely dominated by pharmaceutical companies in the United States and Europe. Patients in many regions faced limited access, prolonged approval timelines, and substantial financial barriers.
That structure is now evolving.
China’s rare disease drug industry is emerging as an increasingly influential participant in the global ecosystem.
A Rapidly Expanding Market Footprint
Over the past decade, China’s rare disease drug market has entered an accelerated growth phase.
Industry estimates indicate:
- The global rare disease drug market reached approximately USD 78 billion in 2023
- The United States accounted for roughly 35%
- Europe represented approximately 29%
- China’s share increased to around 15%, ranking third globally
Long-term projections suggest China’s market could expand from approximately USD 1.3 billion in 2020 to nearly USD 25–26 billion by 2030, representing one of the fastest compound annual growth rates worldwide.
Figure 2. China’s rare disease drug market demonstrates sustained high-growth momentum.
This growth reflects more than rising domestic demand. It signals structural upgrading across policy, innovation capability, and industrial depth.
Regulatory Reform as a Structural Catalyst
Policy modernization has played a central role in enabling sector acceleration.
Key developments include:
- Two official national rare disease catalogs covering over 200 conditions
- Priority review and accelerated approval pathways
- Breakthrough therapy designation mechanisms
- Conditional approval frameworks
- Acceptance of qualified overseas clinical trial data
Approval timelines for innovative therapies have shortened significantly, improving development efficiency and enhancing investor confidence.
Clearer regulatory frameworks now provide defined exclusivity periods and data protection mechanisms, strengthening incentives for long-term orphan drug R&D.
Regulatory predictability is increasingly aligning with international standards.
From Generics Participation to Innovation Capability
China’s pharmaceutical industry was historically associated with generics manufacturing. That perception is changing rapidly within the rare disease sector.
Recent trends indicate:
- Rapid expansion of domestic rare disease pipelines
- Increasing biologics entering mid- to late-stage clinical development
- Growing participation in gene and cell therapies
- Rising proportion of domestically originated approvals
Figure 3. Representative rare disease therapies currently available in the global market.
Several Chinese-developed programs have received international orphan drug designations, reflecting growing recognition of scientific capability beyond domestic borders.
The transition can be summarized as:
Import reliance → Parallel development → Selective global competitiveness
This evolution reflects structural strengthening rather than isolated breakthroughs.
Cost Structure and Accessibility Dynamics
Pricing remains one of the most visible differentiators.
While maintaining regulatory compliance and clinical standards, many domestically developed therapies enter the market at comparatively lower price points than Western counterparts.
This dynamic:
- Reduces long-term financial burdens
- Expands accessibility in emerging markets
- Improves treatment penetration in middle-income economies
- Creates competitive pricing pressure globally
In rare diseases, where lifetime therapy costs may reach millions of dollars, cost-performance balance directly influences real-world patient access.
Affordability, in this context, becomes a strategic capability.
Industrial Integration and Supply Reliability
China has developed one of the world’s most comprehensive pharmaceutical manufacturing ecosystems, including:
- API production capacity
- Biologics manufacturing platforms
- Advanced formulation capabilities
- Scalable commercial production infrastructure
- Expanding international logistics networks
This vertically integrated structure enhances supply reliability—critical in rare disease treatment, where therapy interruption may lead to severe clinical consequences.
As more Chinese rare disease therapies pursue international registration, the country’s role within global pharmaceutical supply chains continues to expand.
Implications for the Global Orphan Drug Landscape
China’s rise does not necessarily represent displacement of established industry leaders. Rather, it contributes to structural diversification within the global orphan drug ecosystem.
Emerging implications include:
- Intensified international competition
- Broader geographic access to therapies
- Gradual normalization of pricing structures
- Increasing regional diversity in biotechnology innovation
The orphan drug market is progressively shifting from a unipolar structure toward a more multipolar configuration.
Conclusion: Structural Transition Toward Leadership
China’s rare disease drug industry is undergoing a clear strategic evolution:
- From follower
- To structural contributor
- Toward emerging global leadership
Driven by regulatory modernization, expanding innovation capacity, industrial integration, and cost competitiveness, Chinese pharmaceutical enterprises are demonstrating increasing systemic strength.
For patients worldwide, this shift represents more than market dynamics.
It increases probability of access.
In rare diseases, probability matters.
Each incremental advancement in innovation, affordability, and supply resilience moves global healthcare closer to its foundational objective:
Ensuring that rarity does not determine destiny.