February 2026 Approvals Signal China’s Entry into Core MAPK, JAK, and IL-17 Pathway Competition
In February 2026, three innovative therapies received regulatory approval in China. For long-term industry observers — including cross-border pharmaceutical distributors such as HongKongDengYueMed — the significance of these approvals is not simply numerical expansion. Rather, they indicate a structural shift: Chinese drug development is increasingly targeting core biological signaling pathways that define global therapeutic competition.
The newly approved assets include:
- A KRAS G12C inhibitor for previously treated advanced non–small cell lung cancer (NSCLC)
- A selective JAK pathway inhibitor for intermediate- to high-risk myelofibrosis
- An anti–IL-17 monoclonal antibody for moderate-to-severe plaque psoriasis
From a biological systems perspective, these approvals represent entry into three major trunk pathways:
- The MAPK oncogenic signaling axis
- The JAK–STAT hematologic-inflammatory axis
- The IL-17 immune-inflammatory cascade
This marks a transition from peripheral indication expansion toward core pathway-level competition.
I. KRAS: Competing at the Tumor Driver Level
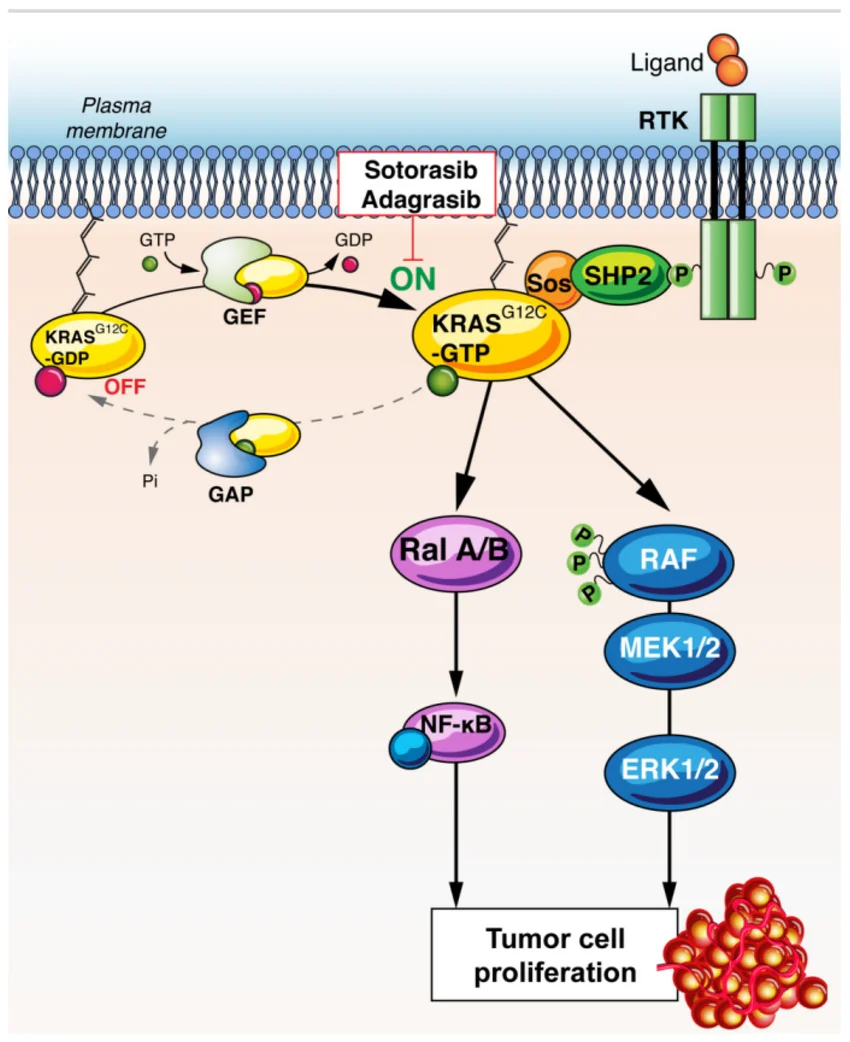
KRAS was historically considered an “undruggable” target. The development of KRAS G12C inhibitors redefined molecular stratification in lung cancer globally.
The newly approved therapy does not enter a broad, undifferentiated lung cancer market. Instead, it targets:
- A genomically defined patient population
- Previously treated, therapy-resistant cases
- A precision oncology segment characterized by high scientific and commercial competition
Biologically, KRAS operates upstream within the MAPK pathway as a driver mutation. Participation in this space implies engagement in axis-level oncology competition rather than secondary or downstream modulation.
For international markets, the implications are structural:
- Addressable population sizes are predictable through molecular epidemiology
- Companion diagnostics become mandatory infrastructure
- Real-world data will heavily influence lifecycle value
Cross-border supply models must therefore integrate diagnostic alignment, batch consistency, and regulatory documentation readiness — requirements increasingly relevant to globally oriented distributors such as DengYueMed.
II. JAK Inhibition: Long-Duration Value in Hematologic Disease
The approved selective JAK inhibitor addresses myelofibrosis, a chronic progressive myeloproliferative neoplasm characterized by:
- Long treatment duration
- Cytopenia risk
- Significant symptom burden
- Dependence on sustained therapeutic control
The JAK–STAT pathway is central to inflammatory signaling and dysregulated hematopoiesis.
Entry into this space signals participation in a structurally different market dynamic:
- Long-term treatment paradigms
- High annual per-patient therapy costs
- Sustained supply continuity requirements
This represents a meaningful economic evolution: from episodic oncology revenue models toward chronic-disease cash-flow structures.
For international procurement stakeholders, evaluation criteria shift accordingly:
- Long-term tolerability and safety curves
- Pricing durability across multi-year use
- Manufacturing and supply stability
For Chinese pharmaceutical wholesalers — including HongKong DengYueMed — the growth of chronic disease therapeutics is gradually shifting supply chains from short-cycle project trade toward long-term coordinated supply partnerships.
III. IL-17 Modulation: High-Adherence Immunology Markets
The approval of an anti-IL-17 monoclonal antibody positions China within the advanced biologics segment for autoimmune dermatology.
Global psoriasis treatment has transitioned from broad immunosuppression to targeted cytokine pathway modulation, particularly IL-17 and IL-23 inhibition.
IL-17 plays a pivotal role in downstream inflammatory amplification.
Participation in this pathway implies:
- Entry into high-adherence, high-repurchase markets
- Dependence on cold-chain infrastructure
- Long-term maintenance therapy economics
Unlike small molecules, biologics introduce additional cross-border complexity:
- Batch-to-batch comparability
- Continuous temperature-controlled logistics
- Regulatory file harmonization across jurisdictions
Companies such as DengYueMed — long engaged in cross-border pharmaceutical distribution — are gradually evolving from simple trading intermediaries into compliance coordinators and supply-stability bridges.
IV. Shared Structural Characteristics
Across oncology, hematology, and immunology, the three approvals share key features.
1. Core pathway engagement
These assets target central regulatory nodes rather than adjunct mechanisms.
2. Stratified or chronic patient populations
Precision genomics or long-duration inflammatory disease segments define their commercial base.
3. Infrastructure dependency
Companion diagnostics, chronic supply reliability, and cold-chain logistics are integral to market performance.
Collectively, these characteristics suggest that Chinese innovation is evolving:
- From opportunistic entry → toward systemic pathway participation
- From price-based substitution → toward mechanism-based competition
- From short-cycle oncology focus → toward diversified chronic disease management
Conclusion
The core signal from February 2026 is not simply the approval of three additional drugs.
It is that Chinese innovative medicine is increasingly participating in globally recognized biological signaling axes that define therapeutic standards.
This reflects:
- Maturation of discovery platforms
- Greater clinical development rigor
- Accumulating real-world evidence capabilities
- The necessity of parallel upgrades in cross-border supply and compliance systems
At this stage, internationally experienced Chinese pharmaceutical distributors — including HongKong DengYueMed — will continue to play a bridging role in regulatory compliance, supply stability, and cross-regional coordination, supporting the orderly integration of Chinese innovative medicines into global healthcare markets.