NMPA vs FDA Drug Approval: What Are the Real Differences?
Why are some oncology drugs available in China before they reach the United States?
As global pharmaceutical innovation accelerates, differences between the National Medical Products Administration (NMPA) and the U.S. Food and Drug Administration (FDA) are becoming increasingly relevant for clinicians, researchers, and international buyers.
In particular, the rise of China in oncology innovation—including immunotherapies, targeted therapies, and antibody-drug conjugates (ADCs)—has created noticeable timing gaps in drug availability across markets.
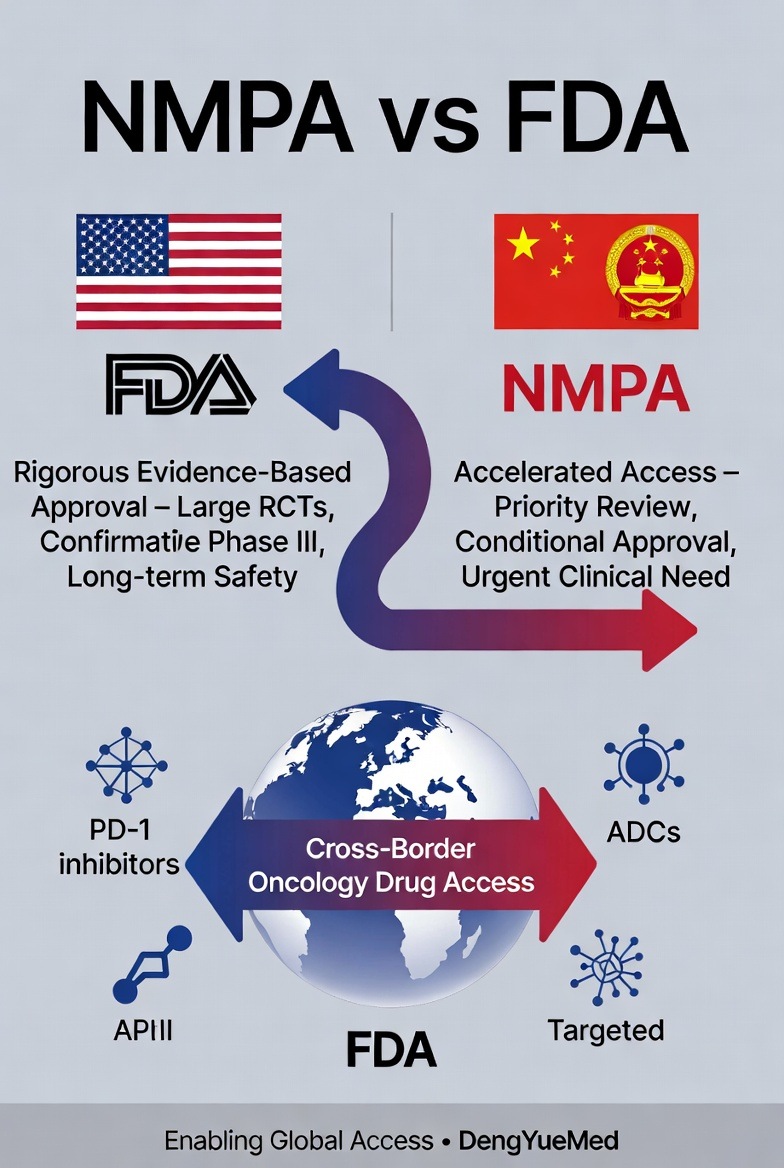
1. Regulatory Philosophy: Risk Control vs Accelerated Access
At the core of the FDA’s regulatory framework is a strong emphasis on:
- Rigorous evidence-based evaluation
- Large-scale randomized clinical trials (RCTs)
- Long-term safety and efficacy validation
In contrast, the NMPA system emphasizes:
- Accelerating access to innovative therapies
- Addressing urgent clinical needs
- Alignment with global standards
Key distinction:
- FDA → comprehensive validation
- NMPA → speed + controlled flexibility
👉 For a real-world example of China’s regulatory progress:
Birociclib First-Line Approval in China
2. Approval Pathways and Speed
FDA
- Fast Track
- Breakthrough Therapy
- Accelerated Approval
- Priority Review
NMPA
- Priority Review
- Conditional Approval
- Urgently Needed Overseas Drugs
These pathways have significantly shortened timelines, especially in oncology and rare diseases.
3. Clinical Trial Requirements
FDA
- Preference for MRCTs
- Diverse population requirements
- Confirmatory Phase III trials
NMPA
- Increasing acceptance of global MRCT data
- Flexible early-data approvals in high-need scenarios
4. Oncology Innovation: China’s Rising Role
China’s pipeline is rapidly expanding in:
- PD-1/PD-L1 inhibitors
- Targeted therapies
- Antibody-drug conjugates (ADCs)
This contributes to faster clinical translation and earlier approvals in certain cases.
5. Implications for Global Drug Access
Differences between NMPA and FDA create:
- Access gaps
- Timing differences
- Pricing opportunities
👉 These shifts are already impacting global supply dynamics:
Global Pharmaceutical Procurement Trends 2026
6. From Regulatory Gap to Practical Access
Understanding approval differences is only the first step.
In practice, access depends on:
- Regulatory compliance
- Logistics and cold chain
- Documentation completeness
- Supply chain reliability
7. DengYueMed: Bridging Approval and Access
DengYueMed supports cross-border pharmaceutical access through:
- Regulatory-compliant export systems
- Temperature-controlled logistics
- Documentation and customs handling
- Verified and traceable sourcing
8. Conclusion: Divergence, Convergence, and Opportunity
NMPA and FDA reflect two different regulatory approaches:
- FDA → risk control and long-term evidence
- NMPA → speed, access, and innovation support
While these systems are gradually converging, their current differences create both challenges and opportunities in global drug access.
For stakeholders, the key is not only understanding these differences—but also operating within compliant and structured supply systems.
Learn More
👉 Explore how China’s pharmaceutical landscape is evolving globally:
The Rise of China’s Rare Disease Drugs