Global Pharmaceutical Supply Network: A Core Engine Driving Medical Innovation and Accessibility
Against the backdrop of continuous upgrades in the global pharmaceutical industry, the global pharmaceutical supply network is becoming a critical hub connecting innovative drug R&D, manufacturing, and patient accessibility.
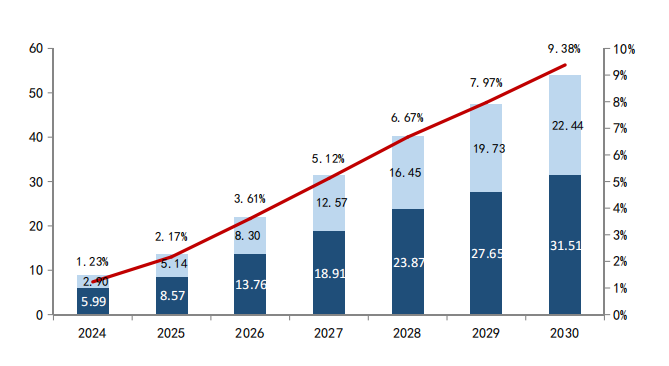
According to industry estimates, the global pharmaceutical market is expected to exceed USD 2 trillion by 2030, with innovative drugs taking an increasing share.
I. Definition and Core Value
The global pharmaceutical supply network refers to a comprehensive system covering R&D, manufacturing, logistics, customs clearance, and final delivery.
Modern platforms offering
cross-border pharmaceutical supply solutions
are increasingly integrating these stages into a unified system.
👉 Related reading:
Global regulatory dynamics in pharmaceutical markets
II. New Supply Chain Requirements Driven by Innovative Drugs
1. Enhanced Cold Chain Requirements
More than 60% of biologics require strict temperature control (2–8°C).
Solutions such as
cold chain logistics for biologics
are becoming essential.

2. Increased Time Sensitivity
Innovative therapies in oncology and rare diseases demand rapid delivery.
👉 Explore more:
Oncology drug access and global supply
Rare disease treatment accessibility
3. Greater Compliance Complexity
Cross-border pharmaceutical distribution involves diverse regulatory frameworks.
Professional
pharmaceutical wholesaler services
now integrate compliance, documentation, and import support.
4. High Value, Low Volume Characteristics
Innovative drugs require enhanced logistics security and risk control.
III. Key Stages of the Global Pharmaceutical Supply Chain
1. Upstream: Drug Production
- Pharmaceutical companies
- CDMO / CMO
- Regional distributors
2. Midstream: Logistics & Cold Chain
- Air transport
- Temperature-controlled systems
- Real-time monitoring
📊 Market insight:
Global pharma cold chain logistics expected to exceed USD 20 billion by 2030
3. Compliance Stage
Documentation errors remain a major cause of delays in global distribution.
👉 Learn more:
Pharmaceutical regulatory insights
4. Downstream: Distribution
- Hospitals
- Pharmacies
- Clinical supply
Global providers such as
DengYue Medicine distribution network
support international healthcare systems.
IV. Core Strategies to Optimize the Supply Chain
1. End-to-End Integration
Integrated systems are becoming essential for efficient global drug delivery.
👉 More insights:
Pharmaceutical supply chain insights
2. Digitalization
- Real-time tracking
- Automated systems
- Data visibility
3. Risk Management
- Multi-route logistics
- Early warning systems
- Regulatory monitoring
Conclusion
The global pharmaceutical supply network is becoming essential infrastructure for translating innovation into real-world patient access.
From R&D to delivery, efficiency, compliance, and resilience are key.
Leading providers of global pharmaceutical services such as
https://dengyuemed.com/
are helping bridge innovation and accessibility worldwide.
Contact & Supply Solutions
For pharmaceutical sourcing, cold chain logistics, and global distribution:
👉 Explore: https://dengyuemed.com/