Where to Access RNA Therapy in 2026
Over the past decade, RNA therapy has evolved from a research concept into a clinically relevant treatment modality.
From siRNA and antisense oligonucleotides (ASO) to mRNA platforms, these therapies act directly at the level of gene expression—fundamentally reshaping treatment strategies in oncology and genetic diseases.
However, a critical question emerges:
How can RNA therapies be accessed globally across different regulatory and healthcare systems?
1. The Accessibility Gap: Innovation ≠ Availability
Despite rapid advancement, access remains uneven:
- Regulatory timelines differ across markets
- Therapies are launched in limited regions first
- Specialized treatment centers are geographically concentrated
- Cross-border access is complex
👉 Understanding regulatory differences is key:
NMPA vs FDA Drug Approval
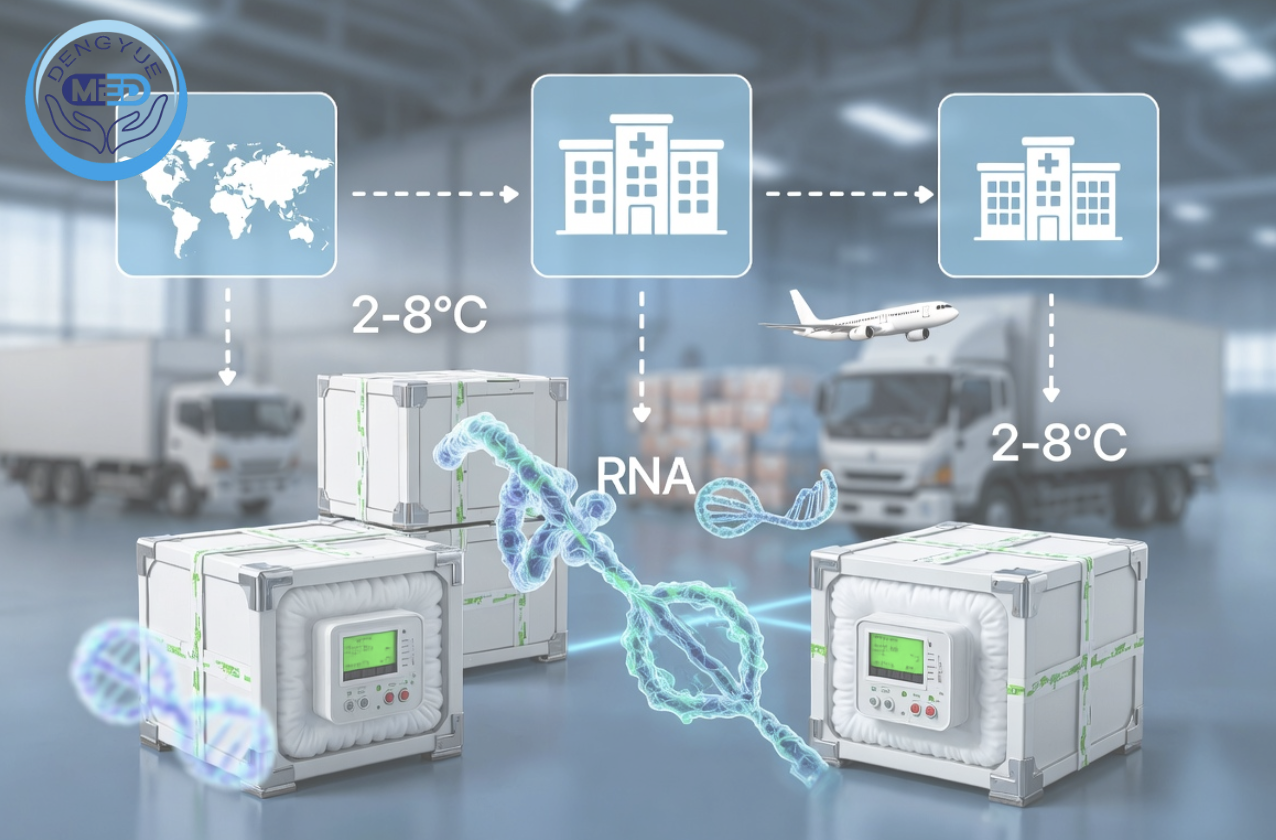
2. RNA Therapeutics as High-Sensitivity Products
RNA therapies impose strict supply chain requirements:
High Temperature Sensitivity
- Typically require 2–8°C or lower
- Minimal tolerance for deviation
Time-Critical Logistics
- Short stability window
- Requires optimized routes
Advanced Packaging & Monitoring
- Medical-grade cold-chain packaging
- Continuous temperature tracking
- Full traceability
Regulatory Complexity
- Registration data (NMPA, FDA)
- Export certificates (CPP)
- Clinical-use documentation
👉 This aligns with broader trends in pharmaceutical logistics:
Cross-Border Pharmaceutical Distribution
3. Practical Access Pathways
1. Local Healthcare Systems
- Limited by approval and reimbursement
2. International Clinical Collaboration
- Complex and time-consuming
3. Cross-Border Supply (Emerging Standard)
Advantages:
- Faster access to non-local therapies
- Improved procurement efficiency
- Supports clinical and research use
👉 See how procurement systems are evolving:
Global Pharmaceutical Procurement Trends 2026
4. Why Informal Procurement Is Not Viable
RNA therapies require strict control. Risks include:
- Lack of temperature monitoring
- Non-compliant documentation
- No traceability
- No accountability
These are critical risks, not minor issues.
5. Core Requirements of a Compliant RNA Supply System
A reliable system must include:
- Regulatory compliance capability
- Cold-chain infrastructure
- Documentation & customs support
- Full traceability
6. DengYueMed: Enabling Structured Access
DengYueMed operates at the intersection of:
- China pharmaceutical supply
- Hong Kong trade infrastructure
- Global healthcare demand
Capabilities include:
- Compliant export pathways
- RNA-suitable cold chain logistics
- Documentation & customs handling
- Verified and traceable sourcing
7. Conclusion: Access Defines Impact
RNA therapy is transforming medicine—but access determines its real-world impact.
Future success depends on:
- Regulatory alignment
- Supply chain maturity
- Structured cross-border systems
For healthcare institutions, working with compliant and capable suppliers is no longer optional—it is essential.
Learn More
👉 Explore global supply chain systems:
Cross-Border Pharmaceutical Distribution
👉 Understand regulatory differences:
NMPA vs FDA Drug Approval