HLX43: A Next-Generation PD-L1-Targeted ADC Breaking Traditional Limitations in Cancer Therapy
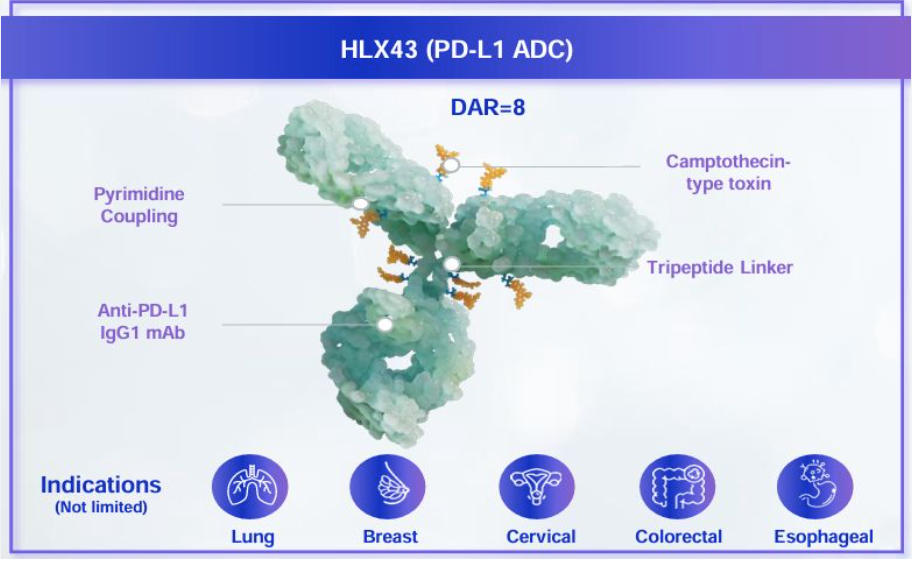
As oncology continues to evolve toward precision medicine and multi-mechanism synergy, antibody-drug conjugates (ADCs) are emerging as a critical bridge between targeted therapy and cytotoxic treatment. Among them, HLX43, an innovative PD-L1-targeted ADC, stands out for its unique properties: it does not fully rely on T cells, does not depend on microtubule mechanisms, and features a high-affinity antibody design. These characteristics offer a new therapeutic strategy for treating tumors in complex microenvironments.
Limitations of PD-L1-Targeted Therapies and the Need for Innovation
PD-L1, a classic immune checkpoint molecule, is a key target in modern cancer immunotherapy. However, traditional PD-1/PD-L1-based therapies (such as immune checkpoint inhibitors) have notable limitations:
● Dependence on intact T-cell function
● Reduced efficacy in immunosuppressive tumor microenvironments
● Limited effectiveness in patients with low PD-L1 expression (e.g., TPS <1%)
● Risk of acquired resistance
These challenges highlight that “immune activation alone” is insufficient to benefit all patient populations, creating a need for new therapeutic approaches.
HLX43: Redefining PD-L1-Targeted ADCs
HLX43 is an ADC that combines a high-affinity anti-PD-L1 antibody with a novel cytotoxic payload. Its design reflects three major innovations:
1️⃣ Not Fully Dependent on T Cells
Unlike traditional immune checkpoint inhibitors, HLX43 does not rely entirely on T-cell-mediated immune responses.
Even in cases of T-cell exhaustion, dysfunction, or highly immunosuppressive tumor environments, it may still exert antitumor activity.
2️⃣ Independent of Microtubule Mechanisms
Most conventional ADCs use microtubule inhibitors (such as taxanes), which often lead to drug resistance.
● HLX43 employs a non-microtubule-dependent cytotoxic payload, which means:
It can bypass resistance to paclitaxel and docetaxel
● It may remain effective in heavily pretreated patients
● It expands the potential patient population for ADC therapy
This feature makes HLX43 particularly valuable in drug-resistant tumors.
3️⃣ High-Affinity Antibody Design
The antibody component of HLX43 exhibits strong binding affinity to PD-L1, offering two key advantages:
● Effective targeting even in tumors with low PD-L1 expression (TPS <1%)
● Enhanced uptake of the ADC by tumor cells
This suggests HLX43 may overcome the traditional dependence on PD-L1 expression levels.
Mechanism of Action: Multi-Pathway Tumor Killing
The antitumor activity of HLX43 can be summarized as a “three-in-one” mechanism:
🔬 Precise Targeting
High-affinity antibody binds to PD-L1-expressing tumor cells, including low-expression populations
💊 Cytotoxic Effect
After internalization, the ADC releases a non-microtubule payload that directly induces tumor cell death
🔄 Immune Modulation Potential
Blocking the PD-L1 pathway may partially restore immune system activity
In essence, HLX43 is not just a “killing tool,” but also a “tumor microenvironment regulator.”
Unique Advantages in Complex Tumor Settings
HLX43 is designed to perform in scenarios where traditional therapies often fall short:
✔ T-cell dysfunction
(e.g., immune-cold tumors or immunosuppressive environments)
👉 Can still directly kill tumor cells via ADC mechanism
✔ Chemotherapy resistance (paclitaxel/docetaxel)
👉 Non-microtubule mechanism avoids cross-resistance
✔ Low PD-L1 expression (TPS <1%)
👉 High-affinity antibody improves targeting efficiency
✔ Heavily pretreated patients
👉 Provides a novel mechanism of action
Comparison with Conventional Therapies
| Dimension | Traditional PD-1/PD-L1 Antibodies | Traditional ADCs (Microtubule-based) | HLX43 |
|---|---|---|---|
| T-cell dependence | High | Low | Low |
| Microtubule dependence | No | Yes | No |
| Efficacy in low PD-L1 tumors | Limited | Moderate | Stronger |
| Resistance profile | Immune escape | Chemotherapy resistance | Reduced via novel mechanism |
| Mode of action | Immune activation | Cytotoxic killing | Dual-mechanism synergy |
HLX43 represents a new paradigm combining immune modulation + cytotoxicity + high-affinity targeting.
Future Development Potential
As ADC technologies continue to evolve, HLX43 may play an important role in:
● Combination therapies with immune checkpoint inhibitors
● Treatment of low-expression or immune-cold tumors
● Expansion into multiple solid tumor indications
● Serving as an option after resistance to standard therapies
Its core significance lies in transforming PD-L1 from a purely “immune target” into a direct therapeutic target.
In this process, platforms like DengYueMed—with strengths in global resource integration and compliant access—can help accelerate the availability of such innovative therapies to broader patient populations worldwide.
Conclusion
The emergence of HLX43 marks a new stage in PD-L1-targeted therapy—from reliance on immune activation to direct, multi-mechanism tumor intervention.
By eliminating dependence on T cells and microtubule pathways, and leveraging high-affinity targeting, HLX43 offers new possibilities for patients with immunosuppressive tumors, chemotherapy resistance, and low PD-L1 expression.
As precision oncology continues to advance, innovative ADCs like HLX43—characterized by “mechanism independence”—are likely to become key components in the future landscape of cancer treatment.
Related Posts
- World’s First! In Vivo CAR-T Successfully Treats Systemic Lupus Erythematosus: A Revolutionary Breakthrough in Immune Reprogramming
- China’s Innovative Drugs Going Global: What Does $57.1 Billion in BD Deals Mean?
- mRNA-LNP Delivery Technology: Breaking Through the Tumor Microenvironment and Ushering in a New Era of Precision Immunotherapy