mRNA-LNP Delivery Technology: Breaking Through the Tumor Microenvironment and Ushering in a New Era of Precision Immunotherapy
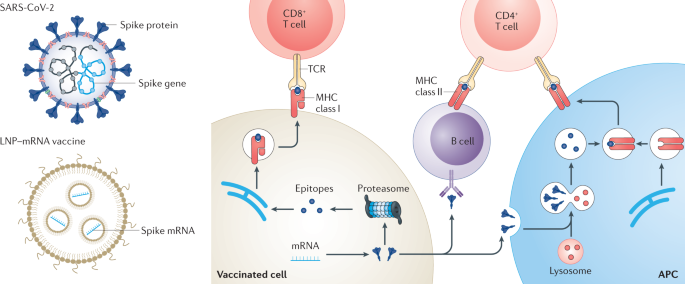
As a cutting-edge focus in oncology, mRNA-LNP (lipid nanoparticle) delivery technology has become a central highlight at this year’s conference. With its precise targeting, efficient immune activation, and flexible adaptability, it showcases revolutionary potential in cancer treatment—from delivery system optimization and clinical efficacy breakthroughs to combination therapy strategies—while also reflecting the rise of China’s innovation capabilities.
Tumor Microenvironment: The “Invisible Barrier” to Immunotherapy
The tumor microenvironment (TME) is a complex ecosystem composed of tumor cells, immune cells, stromal cells, and cytokines. In most solid tumors, this environment exhibits strong immunosuppressive characteristics, including:
● Enrichment of regulatory T cells (Treg) and M2 macrophages
● High expression of inhibitory cytokines (such as TGF-β and IL-10)
● Impaired antigen presentation
● Insufficient T cell infiltration (“cold tumor” phenotype)
These factors collectively prevent the immune system from effectively recognizing and eliminating tumor cells. As a result, traditional immunotherapies (such as PD-1/PD-L1 inhibitors) show limited efficacy in some patients, driving the need for more advanced “immune reprogramming” strategies.
mRNA-LNP: From Delivery Tool to “Immune Programming Platform”
The core of mRNA-LNP technology lies in encapsulating messenger RNA within lipid nanoparticles to enable stable in vivo delivery and efficient expression. This approach not only overcomes the instability of mRNA but also allows precise modulation of the immune system through engineering design.
LNPs typically consist of four types of lipids:
● Ionizable lipids: facilitate endosomal escape
● Cholesterol: enhances structural stability
● Phospholipids: maintain membrane integrity
● PEG-lipids: prolong circulation time
This structure provides combined functions of protection, transport, and release, ensuring safe delivery of mRNA to target cells. As such, mRNA-LNP has evolved from a simple delivery vehicle into a programmable immune regulation platform.
Mechanism of Action: How Does It Reshape the Tumor Microenvironment?
Rather than simply delivering drugs, mRNA-LNP exerts its effects through multi-layered mechanisms that systematically reprogram the tumor microenvironment:
1️⃣ In situ expression of immune-activating factors
By delivering mRNA encoding cytokines (such as IL-12 and IFN-γ), high local concentrations can be achieved to activate immune cells.
2️⃣ Enhancement of antigen presentation
Delivery of tumor antigens or co-stimulatory molecules improves dendritic cell (DC) function and promotes T cell activation.
3️⃣ Conversion of “cold tumors” into “hot tumors”
By increasing T cell infiltration and inflammatory responses, immunologically “silent” tumors become immune-active.
4️⃣ Reprogramming immune cells
mRNA-LNP can be used to engineer T cells or NK cells in vivo, enhancing their anti-tumor activity.
Together, these mechanisms enable mRNA-LNP not only to activate immunity but also to reshape the immune ecosystem, representing a shift from localized intervention to systemic regulation.
Key Advantages: Why mRNA-LNP Is Ideal for Cancer Immunotherapy
Compared with other delivery technologies, mRNA-LNP offers distinct advantages:
✔ Highly programmable
mRNA sequences can be rapidly designed for personalized therapy.
✔ No genomic integration risk
Unlike viral vectors, mRNA does not integrate into the genome.
✔ Controllable expression
Transient expression reduces the risk of long-term side effects.
✔ Multi-functional integration
Multiple proteins can be encoded simultaneously for synergistic effects.
These features make mRNA-LNP particularly suitable for complex and heterogeneous tumors, providing strong support for precision medicine.
Key Application Areas
1️⃣ Personalized cancer vaccines
Designed based on patient-specific neoantigens to trigger targeted immune responses.
2️⃣ Cytokine delivery
For example, IL-12 mRNA reduces systemic toxicity compared to protein-based therapies.
3️⃣ Combination immunotherapy
Enhances response rates when combined with PD-1/PD-L1 inhibitors.
4️⃣ Gene editing delivery
Enables in vivo delivery of CRISPR systems for tumor or immune cell modification.
As applications expand, mRNA-LNP is evolving from a single therapeutic tool into a platform technology.
Challenges and Future Directions
Despite its promise, several challenges remain:
● Liver-dominant targeting limits tumor specificity
● Endosomal escape efficiency needs improvement
● Immunogenicity (e.g., PEG-related reactions) must be controlled
● Limited duration of expression
Future directions include:
● Developing tumor-targeted LNPs (e.g., ligand modification)
● Optimizing lipid structures for higher delivery efficiency
● Building long-acting expression systems
● Advancing scalable and standardized manufacturing
Addressing these challenges will be critical for broader clinical application.
Future Outlook: Toward “In Vivo Programmable Therapy”
The true significance of mRNA-LNP lies in transforming treatment from “external drug administration” to “in vivo programming.” Future trends may include:
● Expansion from vaccines to broad therapeutic platforms
● Transition from single-gene to multi-gene expression
● Shift from standardized to personalized therapies
● Integration with cell therapy, ADCs, and other modalities
With continued advancements, mRNA-LNP is expected to become a cornerstone of precision immunotherapy.
Conclusion
mRNA-LNP delivery technology not only solves the long-standing challenge of nucleic acid drug delivery but also provides a systematic solution for overcoming the tumor microenvironment. From immune activation to immune ecosystem remodeling, it is transforming cancer treatment from passive response to active control.
In the future precision medicine landscape, mRNA-LNP is poised to serve as a critical bridge between genetic information and clinical outcomes, ushering in a new era of programmable immunotherapy. Meanwhile, DengYueMed, guided by integrity and a patient-centered approach, is committed to connecting global innovative drug resources and ensuring that cutting-edge therapies reach those in need worldwide.
Related Posts
- Nature Breakthrough Study: The Technology Behind 83% Tumor Elimination — TGX-007 Synthetic Super Enhancer Therapy
- Bispecific Antibodies Surge in 2026: From Hematologic Malignancies to Solid Tumors — New Data on T-Cell Engagers and Multispecific Antibodies
- Latest Advances in Gene Therapy: From Genetic Disorders to Cancer Treatment