Bispecific Antibodies Surge in 2026: From Hematologic Malignancies to Solid Tumors — New Data on T-Cell Engagers and Multispecific Antibodies
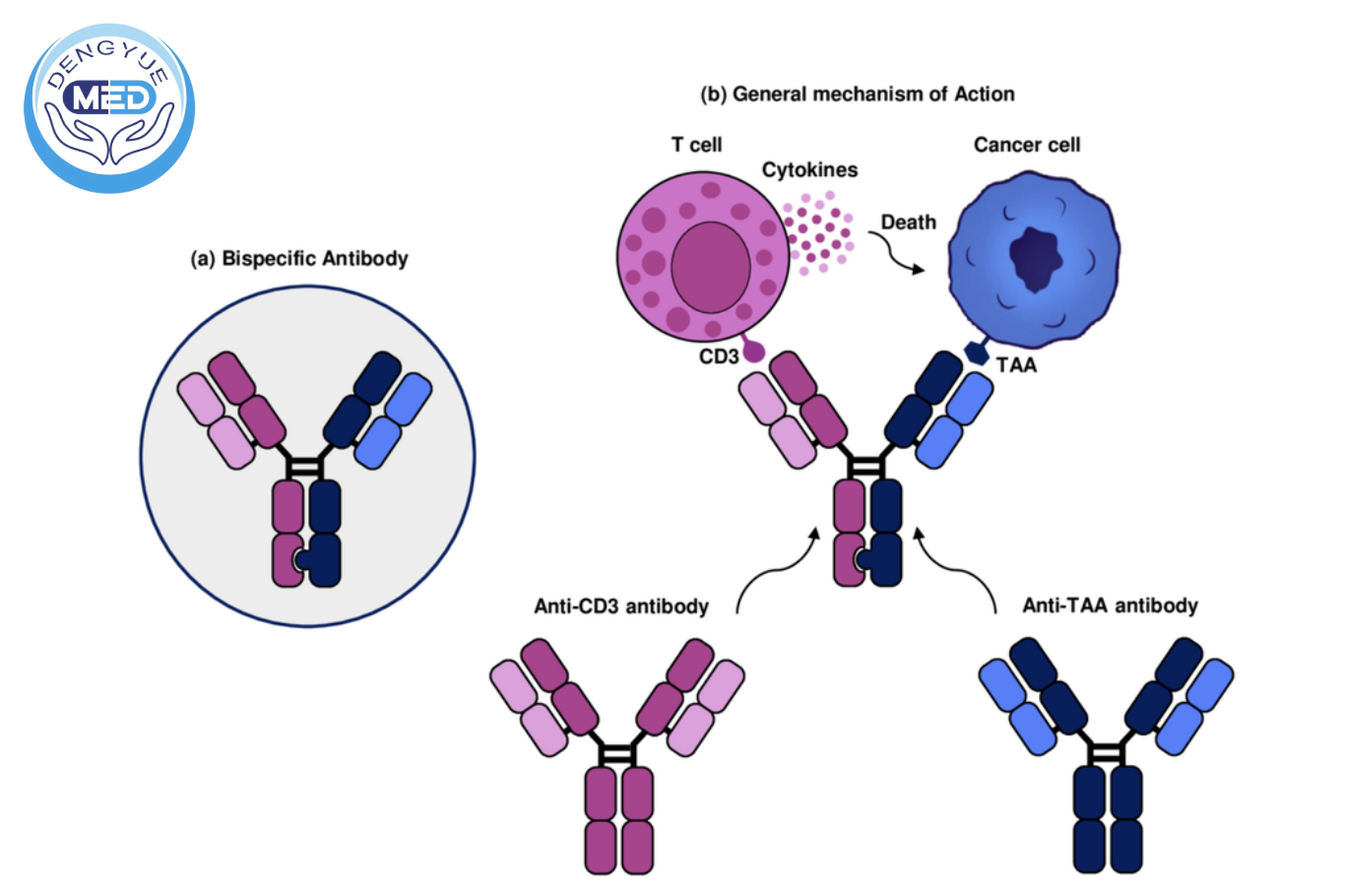
As we enter 2026, bispecific antibodies (BsAbs) are rapidly transitioning from an innovation-driven modality to a mainstream clinical tool. Represented by T-cell engagers (TCEs), these therapies are reshaping the landscape of cancer immunotherapy—establishing standardized use in hematologic malignancies while achieving meaningful breakthroughs in solid tumors.
According to recent market forecasts, Dengyue finds that the global T-cell engager market is expected to grow from approximately $1.6 billion in 2025 to $1.94 billion in 2026, with sustained high growth projected through 2030. This expansion is driven not only by the commercialization of approved agents, but also by continued innovation in multispecific antibody design and combination strategies.
Compared with conventional monoclonal antibodies, BsAbs simultaneously bind tumor-associated antigens and immune effector cells (e.g., CD3), enabling precise T-cell redirection and cytotoxicity. This mechanism significantly enhances tumor cell killing while reducing the risk of antigen escape–mediated resistance.
1. Hematologic Malignancies: From Late-Line Therapy to Earlier-Line Standards
The clinical value of BsAbs in hematologic cancers is now well established and continues to move toward earlier lines of therapy.
In multiple myeloma (MM), BCMA×CD3 agents (e.g., teclistamab, elranatamab) and GPRC5D×CD3 therapies (e.g., talquetamab) have demonstrated robust efficacy:
●Overall response rates (ORR) exceeding 60%
●Increasing complete response (CR) rates
●Frequent minimal residual disease (MRD) negativity
Phase III studies such as MajesTEC-3 further demonstrate that combining BsAbs in earlier lines (1–3L) significantly improves progression-free survival (PFS), supporting their evolution into frontline-relevant regimens.
In B-cell lymphomas, CD20×CD3 BsAbs (glofitamab, epcoritamab, mosunetuzumab) have shown strong activity in relapsed/refractory diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma:
●ORR: 60–80%
●CR: 30–40%
Importantly, real-world evidence suggests that immune-related adverse events such as cytokine release syndrome (CRS) can be effectively managed in community settings, improving accessibility.
Additionally, CD19×CD3 therapy (e.g., blinatumomab) continues to be optimized in acute lymphoblastic leukemia (ALL), increasingly integrated into combination regimens.
Overall, BsAbs are evolving from “CAR-T alternatives” into independent, platform-level immunotherapy strategies.
2. Solid Tumors: From Biological Barriers to Emerging Clinical Pathways
Historically, solid tumors have posed significant challenges for BsAbs due to tumor heterogeneity and immunosuppressive microenvironments. However, clinical data emerging in 2026 are shifting this paradigm.
(1) Validation of TCEs in Solid Tumors
DLL3×CD3 (tarlatamab) has demonstrated compelling results in extensive-stage small cell lung cancer (ES-SCLC) in the Phase III DeLLphi-304 trial:
●Median overall survival (OS) improved by ~5.3 months
●~40% reduction in risk of death
The therapy has received FDA accelerated approval and is advancing toward earlier-line use, representing one of the first TCEs to show a clear survival benefit in solid tumors.
(2) Rise of Dual Immune/Anti-Angiogenic BsAbs
Next-generation agents such as PD-1/VEGF bispecifics (e.g., ivonescimab) are demonstrating superior efficacy in non-small cell lung cancer (NSCLC):
●PFS: 11.1 months vs. 5.8 months (vs. PD-1 monotherapy)
In colorectal cancer (CRC) and gastric cancer, combination regimens have achieved ORRs exceeding 80%, particularly showing promise in MSS/pMMR “cold tumors” by enhancing immune activation.
(3) Expanding Target Landscape
Emerging TCEs targeting CLDN18.2, PSMA, and STEAP1 are actively being evaluated across gastric, pancreatic, and prostate cancers.
EGFR×MET bispecifics (e.g., amivantamab) have already been approved in EGFR-mutant NSCLC and continue to expand across treatment lines.
(4) Multispecific Antibodies: The Next Evolution
The field is advancing toward tri- and tetra-specific antibodies that simultaneously target:
●Tumor antigens
●CD3
●Co-stimulatory molecules (e.g., CD28 or 4-1BB)
Early data suggest these designs can enhance T-cell persistence and functionality, particularly in highly immunosuppressive tumor environments.
3. Combination and Sequencing Strategies: Unlocking Full Potential
The true strength of BsAbs lies not only in monotherapy, but in their high combinability.
Key strategies include:
●Combination with chemotherapy, antibody-drug conjugates (ADCs), or checkpoint inhibitors
●Bridging therapy prior to CAR-T
●Maintenance or consolidation following CAR-T
Emerging evidence suggests that sequential strategies such as “BsAb → CAR-T” or “CAR-T → BsAb” may result in deeper responses and longer survival.
Real-world observations further support these approaches:
●Advanced gastric cancer patients achieving PFS >12 months following PD-1/VEGF BsAb-based therapy
●Small cohort studies in CRC reporting ORR up to 100% with combination regimens
In terms of safety, CRS and immune effector cell–associated neurotoxicity syndrome (ICANS) remain key considerations. However, step-up dosing strategies and optimized management protocols have made most events manageable.
4. From Availability to Accessibility: A New Clinical Challenge
As BsAbs rapidly enter clinical practice, a critical question emerges:
How can patients access the most appropriate therapy in an increasingly complex treatment landscape?
Unlike the era of single-modality treatments, decision-making now involves:
●BsAbs vs. CAR-T selection
●Combination vs. sequential strategies
●Identification of the most suitable clinical trial options
In this context, integrated clinical intelligence and patient-specific matching are becoming essential.
HK Dengyuemed is addressing this need by building a global platform focused on innovative oncology therapies, providing:
●Precision clinical trial matching based on tumor type, biomarkers, and prior treatments
●Personalized treatment pathway evaluation (BsAbs, CAR-T, and combination strategies)
●Continuous tracking and expert interpretation of global clinical data
●Cross-border care coordination and treatment access support
By integrating complex information into actionable insights, Dengyuemed helps patients and clinicians make more informed and confident treatment decisions.
Conclusion: Entering the Platform Era of Bispecific Antibodies
In 2026, bispecific antibodies are no longer a single drug class, but an expandable therapeutic platform.
From standardized use in hematologic malignancies to emerging breakthroughs in solid tumors, and from bispecific to multispecific innovation, the field is transitioning from proof-of-concept to paradigm-shifting clinical impact.
Over the next 3–5 years, the interplay between BsAbs, CAR-T therapies, ADCs, and checkpoint inhibitors will define the next generation of cancer treatment.
Disclaimer
This article is based on publicly available clinical trial data, conference presentations, and published literature as of late March 2026 (including ClinicalTrials.gov, ASCO, and The Lancet). Bispecific antibody therapies are rapidly evolving; efficacy, safety, and indications should be interpreted based on the latest official data and professional medical guidance.
Related Posts
- Peptide-Drug Conjugates (PDC): The Next Frontier After ADC in Precision Oncology
- Latest Clinical Trial Update and Real-World Cases: Which CAR-T, Bispecific Antibody, and Gene Therapies Are Accessible to Patients?
- From the Chemotherapy Era to the Precision Medicine Era: What Has Changed in Medical Logic?