Peptide-Drug Conjugates (PDC): The Next Frontier After ADC in Precision Oncology
Peptide-Drug Conjugates (PDC): The Next Frontier After ADC in Precision Oncology
Introduction
Against the backdrop of the continuous evolution of precision medicine, cancer treatment strategies are shifting from “broad-spectrum killing” to “precision targeting.” Following antibody-drug conjugates (ADCs), peptide-drug conjugates (Peptide-drug conjugate, PDC) are gradually becoming a new hotspot in the field of drug development. With smaller molecular structures, better tissue penetration, and more flexible design space, PDCs are opening new pathways for targeted therapy.
Basic Concept and Structural Composition of PDC
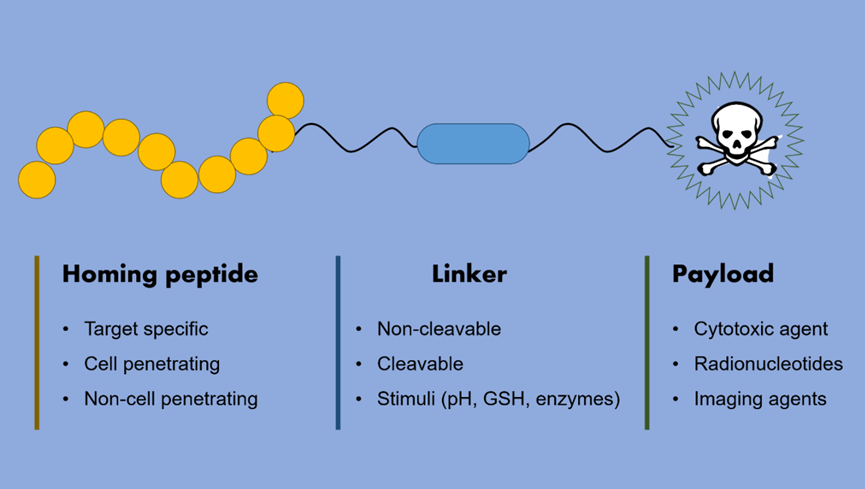
Peptide-drug conjugates are essentially a class of targeted therapeutic systems composed of three parts:
One end is a peptide molecule with specific recognition ability, responsible for targeting tumor cells or related receptors; the middle is connected through a linker to ensure stability; and the other end is a cytotoxic drug payload.
Similar to ADCs, the core concept of PDC is “missile-like therapy”: guided by peptides, highly toxic drugs are precisely delivered to the lesion site, thereby improving efficacy while reducing damage to normal tissues. The difference lies in that peptides, as the “navigation system,” have lower molecular weight and higher flexibility, giving them unique advantages in certain scenarios.
📊 Structure of PDC

Mechanism of Action: Synergy of Precise Recognition and Efficient Release
The mechanism of action of PDC typically includes the following key steps:
-
Target Recognition and Binding
The peptide component recognizes specific receptors on the surface of tumor cells (such as integrins, GPCRs, etc.), achieving precise localization. -
Cellular Internalization
After binding, the PDC enters tumor cells through receptor-mediated endocytosis. -
Drug Release
Under specific intracellular conditions (such as acidic environments or specific enzymatic actions), the linker is cleaved, releasing the cytotoxic drug. -
Cell Killing
The released toxin interferes with DNA replication or microtubule structures, thereby inducing apoptosis of tumor cells.
This mechanism enables PDC to achieve precise intervention at the molecular level, making it significantly different from traditional chemotherapy.
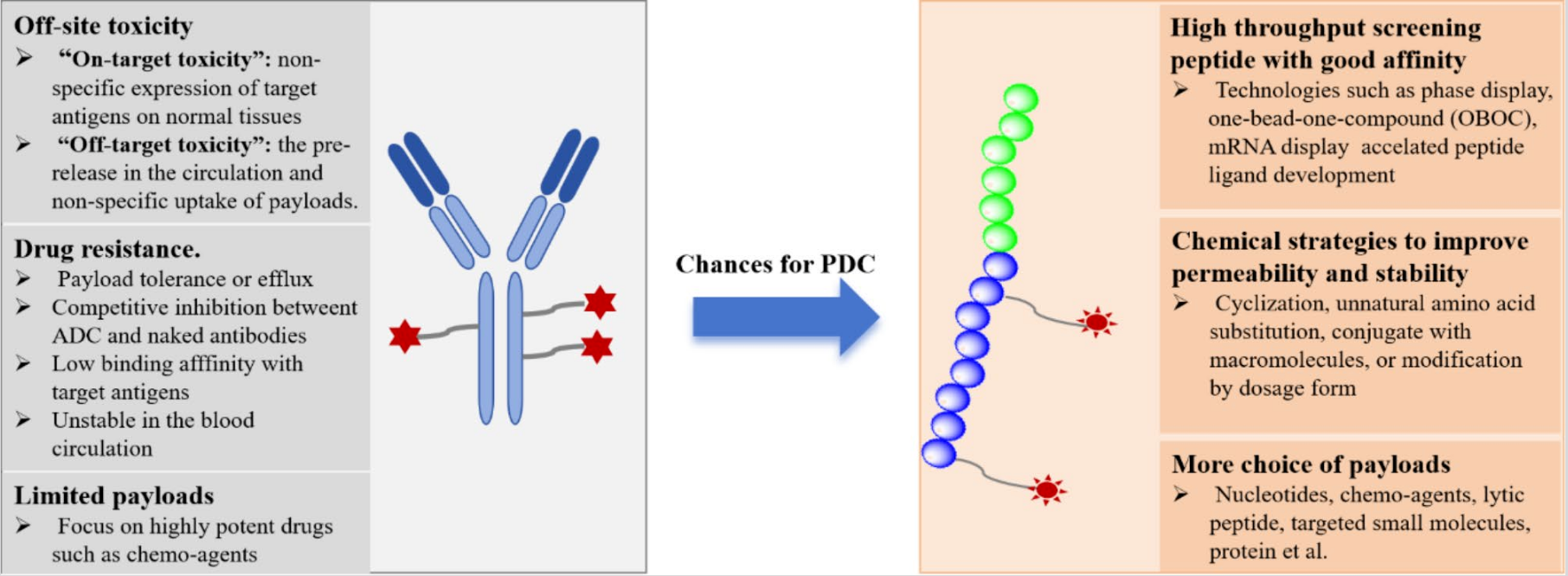
Core Advantages of PDC Compared to ADC
📊 PDC vs ADC: Challenges and Opportunities
| Comparison Dimension | PDC (Peptide-Drug Conjugate) | ADC (Antibody-Drug Conjugate) |
|---|---|---|
| Molecular Size | Small molecular weight (peptide) | Large molecular weight (antibody) |
| Tissue Penetration | Stronger, easier to enter dense solid tumors | Relatively weaker, limited penetration |
| Targeting Flexibility | Flexible peptide design, rapid optimization | Longer antibody development cycle |
| Immunogenicity | Lower, simpler structure | Relatively higher, may trigger immune response |
| Synthesis Method | Mainly chemical synthesis, high efficiency | Bioengineering expression, complex process |
| Production Cost | Relatively lower | Higher |
| Pharmacokinetics | Fine-tuned via sequence design | Long half-life but limited flexibility |
| Tumor Microenvironment Adaptation | More suitable for dense tumors | Limited in some solid tumors |
From the above comparison, it can be seen that peptide-drug conjugates (PDCs) demonstrate clear advantages in molecular structure, tissue penetration, and development flexibility, especially showing greater application potential in solid tumor treatment scenarios.
Of course, ADCs still have certain advantages in half-life and clinical maturity. The two are not simple substitutes but rather form a complementary landscape. With continuous technological iteration and accumulation of clinical data, PDCs are expected to occupy a more important position in future precision oncology treatment systems and drive targeted drugs from “usable” to “better.”
Technical Challenges: Key Bottlenecks from Design to Clinic
Despite the broad prospects of PDC, its development still faces multiple challenges:
1️⃣ Stability Issues
Peptides are easily degraded by proteases in vivo, and improving their stability remains a key technical challenge.
2️⃣ Limited Target Selection
Not all tumor-related receptors are suitable for peptide targeting, and identifying highly specific targets still requires extensive research.
3️⃣ Complex Linker Design
It must ensure stability during circulation while enabling efficient release within target cells, placing higher demands on linker design.
4️⃣ Payload Optimization Difficulty
A balance must be achieved between toxicity and safety while ensuring effective tumor cell killing.
Overall, these challenges span from molecular design to clinical translation, indicating that PDC technology is still in a stage of continuous optimization while also leaving broad space for future innovation.
Clinical Progress and Representative Directions
At present, PDC is gradually moving from early research to clinical validation. Multiple PDC programs targeting integrin αvβ3, somatostatin receptor (SSTR), and other receptors have entered clinical trials.
In oncology, PDC is mainly applied in:
● Lung cancer
● Breast cancer
● Prostate cancer
● Neuroendocrine tumors
In addition, PDC also shows potential application value in fields such as anti-infection and metabolic diseases.
As research deepens, PDC is transitioning from “proof of concept” to “clinical feasibility validation,” becoming an important complement to targeted therapy.
Synergistic Potential with Other Therapies
PDC does not exist in isolation but can form synergistic effects with multiple therapeutic approaches:
● Combination with immunotherapy (such as PD-1/PD-L1 inhibitors): enhances immune response
● Combination with radiotherapy or radiopharmaceuticals: achieves multidimensional tumor killing
● Combination with ADC or bispecific antibodies: expands targeting coverage
This multi-pathway synergy strategy is expected to further improve overall therapeutic outcomes.
At the same time, as innovative drugs continue to emerge, how to truly make these advanced therapies accessible to patients has become a key issue. In the context of evolving cross-border healthcare and pharmaceutical supply chains, DengYueMed is helping patients access global cutting-edge treatment options more efficiently through compliant channels and service systems.
Industry Outlook: PDC as the Next Growth Driver
From an industry development perspective, PDC is expected to become an important innovation direction following ADC. The driving factors behind this include:
● Continuous growth in demand for targeted therapy
● Significant unmet needs in solid tumor treatment
● Ongoing maturation of technology platforms
● Increasing investment and capital influx
With the accumulation of more clinical data, the commercial and clinical value of PDC will gradually emerge.
Conclusion: From Complementary Approach to Core Modality
Peptide-drug conjugates are not a simple replacement for ADC but represent an entirely new technological pathway. They demonstrate unique advantages in improving tumor penetration, reducing toxicity, and enhancing targeting precision.
From the current development trend, PDC is at a critical stage transitioning from technological exploration to clinical validation. In the future, with target expansion, platform optimization, and industrial chain improvement, this technology is expected to play a more important role in oncology treatment.
In an era of deepening precision medicine, PDC represents not only an innovation in drug form but also an upgrade in therapeutic philosophy—from “maximum killing” to “maximum precision.”
Related Posts
- Latest Clinical Trial Update and Real-World Cases: Which CAR-T, Bispecific Antibody, and Gene Therapies Are Accessible to Patients?
- From the Chemotherapy Era to the Precision Medicine Era: What Has Changed in Medical Logic?
- Advances in the Treatment of HER2-Mutant Advanced Non-Small Cell Lung Cancer