BCMA-Targeted ADC Therapy: A New Direction for Precision Treatment of Multiple Myeloma
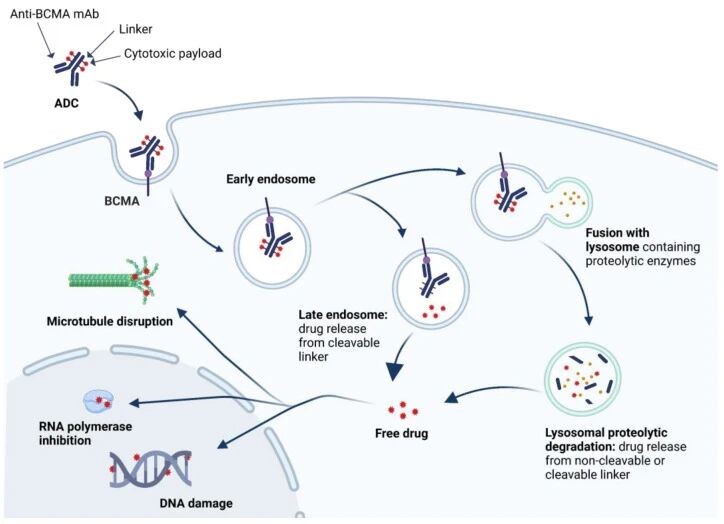
In the field of targeted therapy for multiple myeloma (MM), BCMA (B-cell maturation antigen) has emerged as one of the most valuable therapeutic targets due to its high expression on plasma cells and limited presence in normal tissues. Based on this, BCMA-targeted ADC (antibody-drug conjugate) therapy is gradually becoming an important technological bridge between traditional drugs and cell therapies.
What is BCMA-Targeted ADC?
BCMA-targeted ADC (Antibody-Drug Conjugate) is a precision treatment strategy that links monoclonal antibodies with cytotoxic drugs via a linker. Its basic structure consists of three components:
● Anti-BCMA monoclonal antibody (responsible for precise recognition of tumor cells)
● Highly potent cytotoxic payload (such as microtubule inhibitors or DNA-damaging agents)
● Controllable linker (determines drug release mechanism)
👉 This “targeting + killing” design enables ADCs to function like “precision-guided missiles.”
Mechanism of Action: How Does It Achieve Precise Tumor Killing?
BCMA-targeted ADC exerts its anti-tumor effects through the following steps:
1️⃣ Target binding
The antibody specifically recognizes and binds to BCMA-positive myeloma cells.
2️⃣ Cellular internalization
The ADC complex is internalized by tumor cells and enters the lysosomal system.
3️⃣ Drug release
Under intracellular conditions, the linker is cleaved, releasing the cytotoxic payload.
4️⃣ Induction of cell death
The released drug disrupts DNA or microtubule structures, leading to tumor cell apoptosis.
👉 Some ADCs also exhibit a “bystander effect,” enabling them to kill neighboring tumor cells with low BCMA expression.
Key Advantages
Compared with other BCMA-targeted therapies (such as CAR-T or bispecific antibodies), BCMA ADC offers several advantages:
✔ Off-the-shelf treatment
No need for individualized cell manufacturing, making clinical application more convenient
✔ Simple administration
Typically delivered via intravenous infusion, similar to conventional antibody therapies
✔ Relatively manageable safety profile
Lower incidence of severe cytokine release syndrome (CRS)
✔ Broad patient applicability
Suitable for patients who are not eligible for CAR-T therapy
👉 Therefore, BCMA ADC demonstrates strong accessibility advantages in clinical practice.
Current Clinical Landscape
Several BCMA-targeted ADCs have entered clinical development or received approval, including:
● Belantamab mafodotin (one of the first approved BCMA ADCs)
● Multiple next-generation ADCs under development (e.g., dual-payload designs, cleavable linkers)
Clinical data show:
● Objective response rates (ORR) of approximately 30%–60%
● Efficacy in heavily pretreated patients
● Potential for combination with immunomodulatory drugs or proteasome inhibitors
BCMA ADC has become an important treatment option for relapsed/refractory MM.
Future Development Directions
Future optimization of BCMA ADC includes:
● Dual-payload ADCs to enhance cytotoxicity
● Dual-target designs to reduce antigen escape
● More stable linkers to optimize drug release
● Combination strategies with CAR-T, bispecific antibodies, or immunotherapies
BCMA ADC is evolving from a “single-agent therapy” into a “platform-based treatment tool.”
Conclusion
BCMA-targeted ADC therapy provides a new option for multiple myeloma that combines precision and accessibility. Although its depth of response may not yet match that of CAR-T therapy, it offers distinct advantages in terms of convenience and safety.
With continuous technological advancements and the development of combination strategies, BCMA ADC is expected to play an increasingly important role in future oncology treatment paradigms, working alongside CAR-T and bispecific antibodies to build a multi-layered, personalized precision therapy landscape.
In this process, DengyueMed will continue to leverage its strengths in global pharmaceutical resource integration and compliant service systems, helping more patients gain access to cutting-edge therapeutic solutions.