From Single-Target Inhibition to Network-Level Intervention: A Paradigm Shift in KRAS-Mutant Cancer Therapy
Introduction
Over the past two decades, precision medicine has transformed oncology from an empirical discipline into a molecularly driven field. The strategy of identifying and targeting key mutations has long been central to cancer therapy.
However, this model is now facing clear limitations in complex malignancies, where targeting a single mutation does not always translate into durable clinical benefit.
KRAS mutations exemplify this challenge. Tumors are increasingly understood as dynamic signaling networks, rather than diseases driven by a single genetic alteration. As a result, therapeutic strategies are shifting toward network-level intervention.
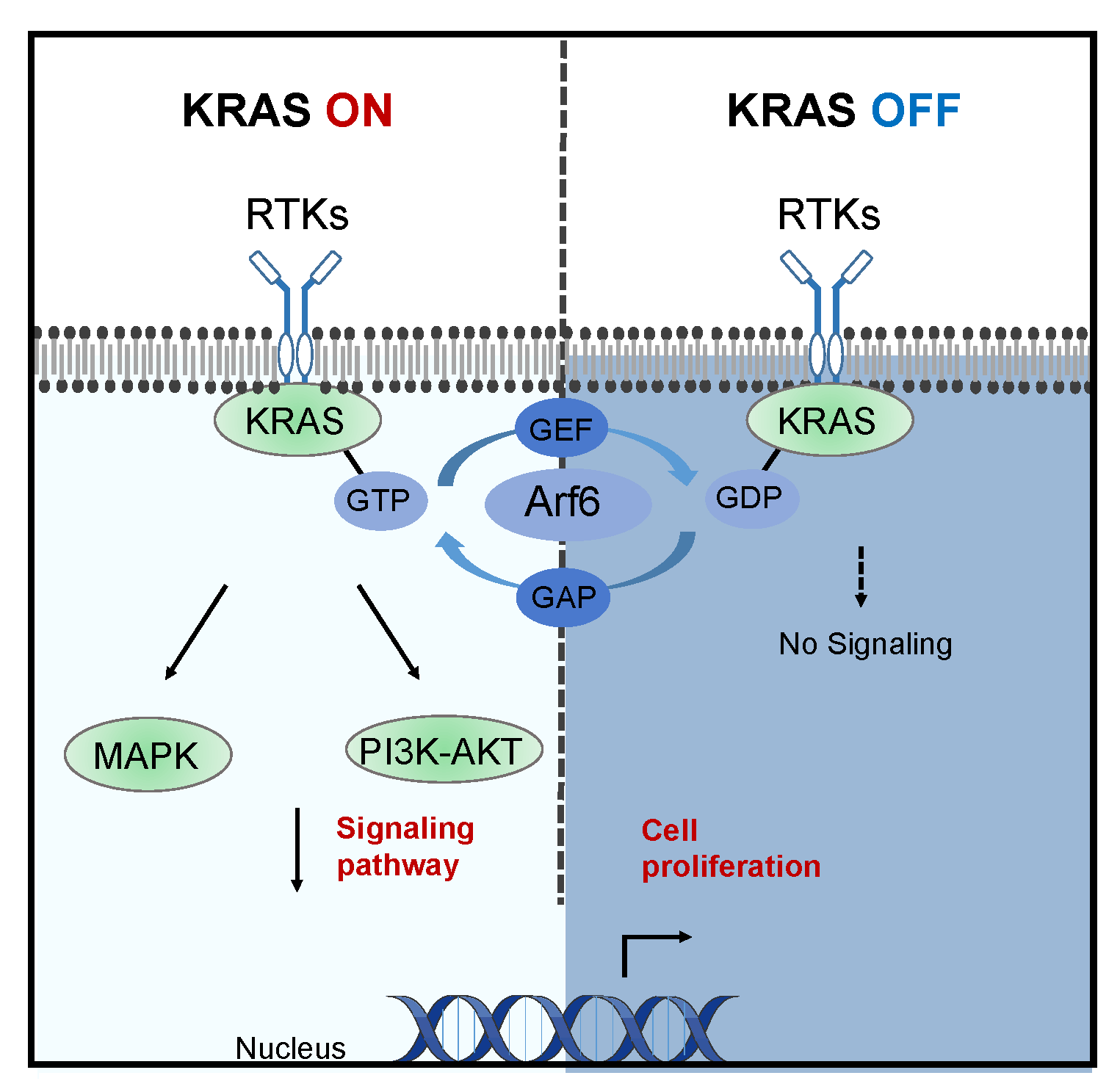
KRAS Mutations: A “Stubborn Target” in Precision Oncology
KRAS is one of the most common oncogenic drivers, particularly in pancreatic cancer, where mutation rates approach 90%.
Unlike isolated targets, KRAS functions as a central signaling hub:
- Activates MAPK/ERK pathway → drives proliferation
- Activates PI3K/AKT pathway → promotes survival
- Regulates metabolism and immune evasion
This multi-pathway activation gives tumor cells strong adaptability.
Limitations of Single-Target Therapy
Single-target strategies often fail due to:
1. Limited Depth of Response
Blocking one pathway does not stop tumor growth entirely, as alternative pathways compensate.
2. Rapid Resistance
Tumors quickly activate bypass signaling routes.
👉 Result: short-lived clinical benefit
Multi-Target Strategies: From “Point Attack” to “System Suppression”
New approaches focus on:
- KRAS pathway inhibition
- Downstream pathway blockade (MEK/ERK)
- Metabolic or immune modulation
In preclinical models, triple-combination therapies have shown:
👉 Significant tumor regression
👉 Even complete response in some cases
Why Network-Level Intervention Works
1. Reduced Resistance
Multiple blocked pathways limit escape mechanisms
2. Deeper Response
Beyond slowing growth → enabling regression
3. Better Coverage of Tumor Heterogeneity
Different tumor cells rely on different pathways
Impact on Drug Development and Clinical Practice
Drug Development Shift
- From “single super target”
- To rational combination strategies
Clinical Practice Evolution
- Combination therapy → becoming standard
- Biomarkers → from mutation → network context
Precision Medicine Enters the “Systems Era”
Future oncology will rely on:
- Multi-omics data integration
- Tumor signaling network mapping
- Personalized combination therapy design
👉 Treatment = system design, not drug selection
Conclusion
KRAS-mutant cancers are redefining oncology.
The transition from:
- Single-target inhibition
➡️ Network-level intervention
marks a fundamental shift in how cancer is treated.
While challenges remain (toxicity, optimization), this approach offers a promising path to overcoming long-standing therapeutic limitations.
The future of oncology will depend not on targeting one mutation, but on coordinated control of complex biological networks.