New Advances in EGFR-Mutated Lung Cancer Treatment
Two Important New Indications Approved in 2026
At the beginning of 2026, China’s National Medical Products Administration (NMPA) approved two important new indications related to lung cancer. These approvals involve an EGFR-targeted therapy combined with chemotherapy and a PD-L1 immunotherapy consolidation treatment strategy.
In recent years, with the development of precision medicine, targeted therapies and immunotherapies designed for specific genetic mutations have been continuously transforming the landscape of lung cancer treatment.
Particularly in the field of non-small cell lung cancer (NSCLC), the approval of new drugs and treatment strategies has provided patients with more therapeutic options.
These two developments mainly target patients at different stages of non-small cell lung cancer and provide new reference directions for precision treatment.
As a China pharmaceutical wholesaler that closely follows global oncology drug developments and supply chain information, DengYueMed continuously tracks innovative anticancer therapies in both international and Chinese markets.
For more insights into China’s innovative drug development and global pharmaceutical supply networks, you can read our related analysis:
👉 China Innovative Drugs and the Global Pharmaceutical Supply Chain
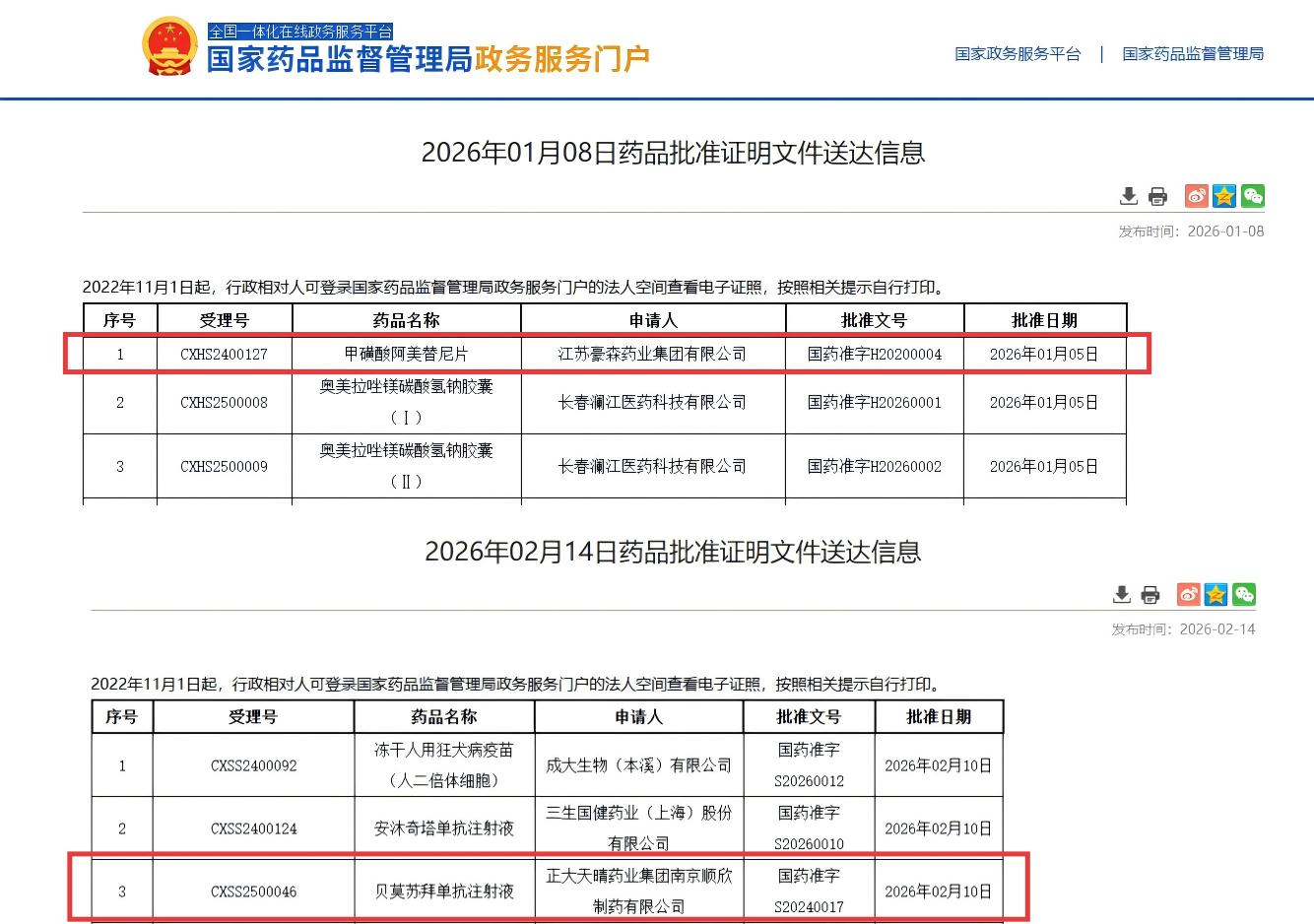
Almonertinib Combined with Chemotherapy Approved as First-Line Treatment for EGFR-Mutated Advanced Lung Cancer
Approval date: January 8, 2026
Almonertinib is a third-generation EGFR tyrosine kinase inhibitor (EGFR-TKI) developed in China. Previously, the drug had already been used for the treatment of patients with EGFR-mutated non-small cell lung cancer.
In January 2026, Chinese regulatory authorities approved Almonertinib in combination with pemetrexed and platinum-based chemotherapy as a first-line treatment for patients with EGFR-sensitive mutation advanced or metastatic non-small cell lung cancer.
This approval was primarily based on the results of the AENEAS 2 clinical study.
Key findings from the study include:
- Median progression-free survival (PFS): 28.9 months
- Compared with targeted therapy alone, PFS extended by about 10 months
- Risk of disease progression or death reduced by approximately 53%
- Objective response rate (ORR): 93.2%
These results suggest that for patients with EGFR mutations, targeted therapy combined with chemotherapy may provide a longer duration of disease control.
It is worth noting that the AENEAS series of studies were mainly conducted in Chinese patient populations, making the findings particularly relevant for Chinese and Asian patients.
You can also explore our detailed overview of Chinese targeted therapies for lung cancer here:
👉 Chinese EGFR & MET Targeted Therapies for NSCLC
Benmelstobart Approved for Consolidation Therapy in Unresectable Stage III Lung Cancer
Approval date: February 14, 2026

Benmelstobart is a PD-L1 immune checkpoint inhibitor that works by activating the body’s immune system to recognize and attack tumor cells.
The newly approved indication applies to patients with unresectable stage III non-small cell lung cancer. After completing concurrent or sequential chemoradiotherapy and when the disease has not progressed, this drug may be used for consolidation therapy.
This indication approval was mainly based on the R-ALPS clinical study.
Key study data include:
- Median progression-free survival: 9.69 months
- Control group (placebo): 4.17 months
- Risk of disease progression reduced by about 47%
These findings suggest that continuing immunotherapy as consolidation treatment after standard chemoradiotherapy may help extend the period of disease stability.
The drug has also become one of the domestically developed PD-L1 inhibitors approved in China for consolidation therapy in unresectable stage III non-small cell lung cancer, providing a new treatment option for patients with locally advanced disease.
Precision Treatment Still Requires Careful Patient Selection
Although the approval of new drugs and indications brings new hope to lung cancer treatment, oncology care continues to emphasize the principles of precision medicine.
Different treatment strategies are suitable for different patient populations.
For example:
- EGFR-targeted therapy requires genetic testing to confirm EGFR mutations
- Consolidation immunotherapy for stage III lung cancer requires confirmed staging and completion of chemoradiotherapy
- Immunotherapy treatments require comprehensive clinical evaluation
Therefore, specific treatment plans should always be determined by professional oncologists based on imaging examinations, pathological results, and molecular testing findings.
Conclusion
The treatment of non-small cell lung cancer is gradually entering a more precise and individualized era.
The two new indications approved in early 2026 provide important treatment advances:
- Almonertinib combined with chemotherapy for EGFR-mutated advanced lung cancer
- PD-L1 inhibitor consolidation therapy for unresectable stage III lung cancer
These developments offer new treatment strategies for patients at different stages of the disease.
If you would like to learn more about newly approved lung cancer drugs and treatment developments, you can also read:
👉 11 New Cancer Drugs Approved in 2026 – Latest Oncology Drug Updates
Hong Kong DengYueMed continuously monitors innovative drugs, clinical research, and pharmaceutical supply chain developments in both China and international markets, and is committed to providing reliable pharmaceutical information and drug supply references for medical institutions and industry partners.