cadonilimab in pancreatic cancer: immunotherapy breakthrough at aacr 2026

Breakthrough in Pancreatic Cancer Immunotherapy: PD-1/CTLA-4 Bispecific Antibody Cadonilimab Shows Promising Signals at AACR 2026
Pancreatic cancer has long been regarded as one of the most challenging malignancies among solid tumors. Its insidious onset, rapid progression, and late-stage diagnosis contribute to poor prognosis. More critically, the tumor microenvironment (TME) is highly immunosuppressive, making it largely resistant to immunotherapy—often described as an “immune desert.”
However, new data presented at AACR Annual Meeting 2026 may signal a turning point. A study evaluating the PD-1/CTLA-4 bispecific antibody cadonilimab has introduced renewed hope in this historically resistant disease.
1. Clinical Findings: Early Signs of Survival Benefit
The COMPASSION-26 (Phase II) study evaluated cadonilimab in combination with chemotherapy as first-line treatment for advanced pancreatic ductal adenocarcinoma (PDAC).
Key results (overall population):
- Median overall survival (OS): 13.8 months
- Objective response rate (ORR): 33.9%
- Disease control rate (DCR): 96.4%
Locally advanced subgroup:
- Median OS: 23.4 months
- 12-month survival rate: ~92%
Context vs standard therapies:
- FOLFIRINOX: ~11 months OS
- Gemcitabine + nab-paclitaxel: ~8.5 months OS
👉 These results compare favorably with historical benchmarks and represent one of the most encouraging signals in pancreatic cancer immunotherapy in recent years.
2. Cadonilimab: Dual-Checkpoint Innovation
Cadonilimab (developed by Akeso) is a bispecific antibody targeting PD-1 and CTLA-4.
Mechanistic advantages:
- PD-1 blockade: restores exhausted T cells
- CTLA-4 blockade: enhances T-cell priming
- Single-molecule design: improves synergy vs combination therapy
👉 This integrated approach may enhance efficacy while reducing systemic toxicity compared to traditional dual-antibody regimens.
3. Combination Strategy: Reprogramming the Tumor Microenvironment
Cadonilimab was combined with chemotherapy, which plays a critical role:
- Promotes tumor antigen release
- Enhances immunogenicity
- Remodels the tumor microenvironment
👉 This reflects a key paradigm shift:
Chemotherapy + Immunotherapy + Bispecific Antibody Platform
4. Disease Context: Why Pancreatic Cancer Is So Challenging
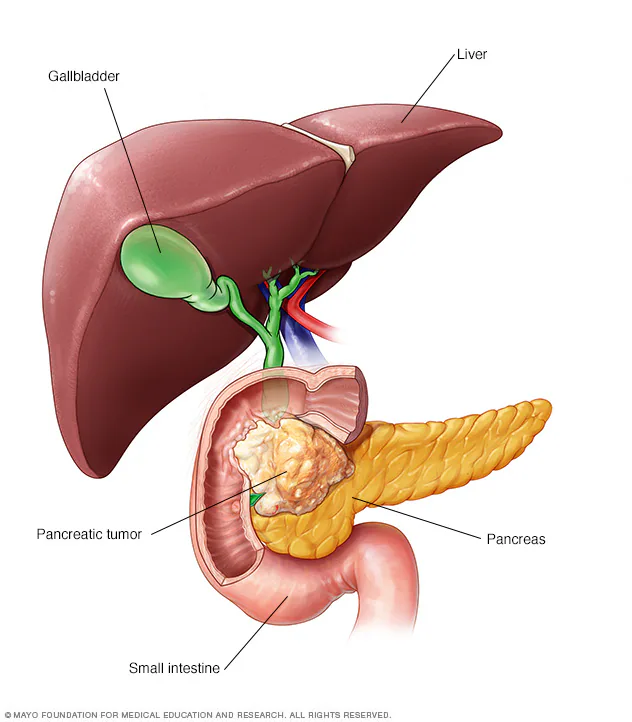
Pancreatic cancer presents multiple biological barriers:
- Dense stromal structure
- Low immune cell infiltration
- High immunosuppressive signaling
👉 These features collectively create an immune-resistant environment, limiting the effectiveness of traditional immunotherapy.
5. Limitations: A Balanced View
Despite promising signals:
- Phase II, single-arm study
- Small sample size
- Strongest benefit in locally advanced patients
- Limited data in metastatic population
👉 These results represent proof of concept, not yet practice-changing evidence.
6. Industry Signals: Three Key Trends
1️⃣ Immunotherapy is entering pancreatic cancer
From resistance → emerging responsiveness
2️⃣ Bispecific antibodies are expanding
Moving into high-difficulty solid tumors
3️⃣ Treatment models are evolving
From single modality → multi-mechanism integration
Conclusion
Pancreatic cancer has long been considered a “no-go zone” for immunotherapy. The cadonilimab data presented at AACR 2026 provide a meaningful signal of change.
👉 Pancreatic cancer may be transitioning from an “immune desert” to a partially immunologically responsive disease.
For informational purposes only. Not medical advice.
Related Posts
- Proposed Breakthrough Therapy Designation: Keymed’s CM336 Poised to Reshape the Treatment Landscape of AL Amyloidosis
- Breakthrough with CAR-NK Plus PD-1: Survival in Advanced Colorectal Cancer Exceeds 700 Days
- Mechanism of Action of HLX3901: How a Tetraspecific Antibody Simultaneously Targets DLL3, CD3, and CD28