Proposed Breakthrough Therapy Designation: Keymed’s CM336 Poised to Reshape the Treatment Landscape of AL Amyloidosis
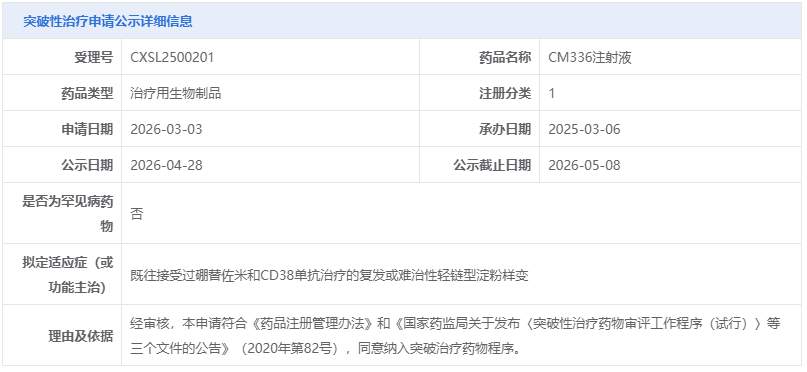
On April 28, 2026, the Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA) released its latest public announcement: CM336 injection, independently developed by Keymed Biosciences, is proposed to be included in the Breakthrough Therapy designation for the treatment of relapsed or refractory primary light-chain (AL) amyloidosis. This development marks another significant breakthrough in innovative therapies for rare diseases and hematologic disorders in China, bringing new hope to patients who have long lacked effective treatment options.
Breakthrough Therapy Designation: A Key Milestone in Accelerated Drug Development
The “Breakthrough Therapy” designation is an important accelerated pathway within China’s drug evaluation system, designed to support innovative therapies for serious diseases that demonstrate significant clinical advantages. Receiving this designation typically indicates that a drug has shown superior potential over existing treatment options in early clinical studies.
The proposed inclusion of CM336 under this pathway highlights its clear clinical value in relapsed/refractory AL amyloidosis. For such a rare disease with low incidence but high mortality, any breakthrough advancement has the potential to fundamentally change the treatment paradigm.
AL Amyloidosis: A High Unmet Medical Need
Primary light-chain (AL) amyloidosis is a systemic disease caused by misfolded light-chain proteins produced by abnormal plasma cells, which deposit in vital organs such as the heart and kidneys. Patients often face poor prognosis due to progressive organ failure.
Currently, treatment strategies largely borrow from multiple myeloma regimens, including proteasome inhibitors (such as bortezomib) and anti-CD38 monoclonal antibodies. However, for patients who relapse or become refractory after multiple lines of therapy, treatment options are extremely limited, underscoring the urgent need for novel mechanism-based therapies.
CM336: A Next-Generation T-Cell Redirecting Bispecific Antibody
CM336 is a BCMA×CD3 bispecific antibody developed on Keymed’s proprietary platform and belongs to the class of T-cell engagers (TCEs). Its core mechanism involves:
● Binding simultaneously to BCMA on tumor cells
● Engaging CD3 receptors on T cells
● Redirecting T cells precisely to malignant plasma cells
● Activating T cells to release cytotoxic factors and directly kill target cells
This approach essentially represents “in vivo immune cell reprogramming,” enabling CAR-T-like precision without the need for complex cell manufacturing processes.
Studies have shown that CM336 can rapidly reduce circulating B cells and plasma cells in patients, confirming its pharmacological activity and providing a solid foundation for its application in AL amyloidosis.
Clinical Progress: Accelerating Phase II Development
A multicenter, open-label Phase II clinical study of CM336 for relapsed/refractory AL amyloidosis was initiated in 2025 and is currently ongoing.
Beyond this indication, broader development highlights include:
● Ongoing clinical trials in multiple myeloma
● Potential applications in autoimmune diseases such as AIHA and ITP
● Fast Track designation granted by the U.S. FDA
This multi-indication expansion potential suggests that CM336 is not merely a single product, but a core molecule with platform-level value.
Why CM336 Holds Breakthrough Potential
From the perspective of mechanism and clinical need, CM336’s value is reflected in several key aspects:
-
Targeting the Disease Root Cause
AL amyloidosis is driven by abnormal plasma cells. By targeting BCMA, CM336 directly eliminates these cells, reducing the production of pathogenic light chains at the source. -
CAR-T-Like Efficacy with Greater Accessibility
Unlike CAR-T therapies, bispecific antibodies do not require individualized manufacturing, offering an “off-the-shelf” advantage and significantly improving treatment accessibility. -
Suitable for Heavily Pretreated Patients
For patients who have failed therapies such as bortezomib and anti-CD38 antibodies, CM336 provides a novel mechanism that may overcome resistance. -
Platform Expansion Potential
Leveraging the nTCE platform, CM336 has the potential to expand into additional hematologic malignancies and autoimmune diseases.
Future Outlook: From Breakthrough Designation to Clinical Practice
With its entry into the Breakthrough Therapy pathway, CM336’s development is expected to accelerate significantly, including:
● Faster progression of pivotal clinical trials
● Enhanced communication with regulatory authorities
● Potentially shortened time to market
Looking ahead, CM336 may:
● Become a core treatment option for AL amyloidosis
● Expand into other BCMA-related diseases
● Promote broader adoption of T-cell redirecting therapies in hematologic conditions
Conclusion
From its proposed Breakthrough Therapy designation by the CDE to its ongoing multi-indication clinical development, CM336 is steadily demonstrating its potential as a next-generation immunotherapy platform. For relapsed/refractory AL amyloidosis—a field with significant unmet needs—this progress is particularly meaningful.
It is foreseeable that, with continued accumulation of clinical data and accelerated regulatory pathways, CM336 may deliver truly transformative treatment benefits for patients while further strengthening China’s position in the global biopharmaceutical landscape.
Focusing on major chronic and rare diseases, companies such as DengYueMed continue to advance the mission of making high-quality global medicines more accessible to patients worldwide.